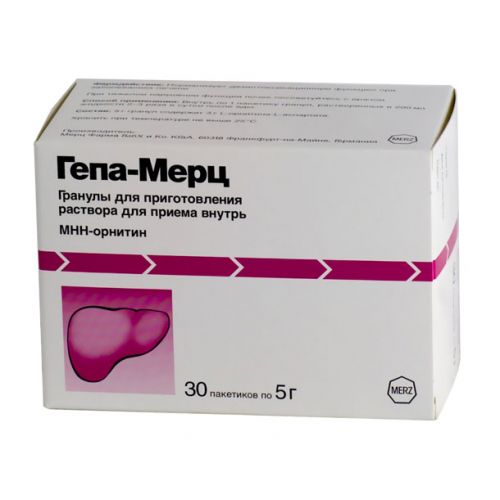
Hepa-Merz 5g 30s granules for oral solution
$134.50
Description
The instruction for medical use of medicine Gepa-Mertz the Trade name Gepa-Mertz the International unlicensed name is not present the Dosage form Granulate for preparation of solution for intake, 5 g Structure 1 bag active agent – L-ornitina-L-contains aspartate of 3.0000 g, excipients: anhydrous citric acid, sodium saccharin, sodium cyclomate, fragrance lemon, fragrance orange, yellow sunset of FCF (E110), povidone 25, fructose the Description of the Granule from light orange till orange color, loose, with a citrus smell. 5% solution of drug of light orange color, slightly opalescent. Pharmacotherapeutic group Digestive tract and metabolism. Drugs for treatment of diseases of a liver and biliary tract. Drugs for treatment of diseases of a liver. The ATX A05BA code the Pharmacological Ornithine Pharmacokinetics properties aspartate dissociates on the components making it – amino acids ornithine and aspartate which are soaked up in a small intestine by active transport through an intestinal epithelium. Both amino acids have short elimination half-life 0.3-0.4 hours. It is removed with urine through a mochevinoobrazovaniye cycle. In vivo pharmacodynamics action of L – ornithine – L – aspartate is carried out by means of two key methods of a detoxication of ammonia: synthesis of urea and synthesis of glutamine by means of amino acids – ornithine and aspartate. Synthesis of urea happens in periportal hepatocytes where ornithine acts as the activator of two enzymes: ornitinkarbamoiltransferaza and karbamoilfosfatsintetaza and also as substrate for urea synthesis. Synthesis of glutamine happens in perivenous hepatocytes. Under pathological conditions aspartate and other dicarboxylates, including ornithine metabolism products, are absorbed in cells and used in the form of glutamine for ammonia binding there. The glutamate serves as the amino acid connecting ammonia as in physiological, and pathological conditions. The received amino acid represents glutamine not only a non-toxic form for ammonia removal, but also activates an important cycle of ureapoiesis (intracellular exchange of glutamine). In physiological conditions ornithine and aspartate do not oppress urea synthesis. Drug reduces the increased ammonia level in an organism, in particular, in liver diseases. Effect of drug is connected with its participation in an ornitinovy tricarbonic acid cycle (ureapoiesis from ammonia). Promotes production of insulin and somatotropic hormone. Improves protein metabolism in the diseases demanding parenteral nutrition. Indications – the acute and chronic diseases of a liver accompanied with a giperammoniyemiya (including cirrhoses, a viral hepatitis, in fat dystrophy, toxic damages of a liver against the background of an alcoholic poisoning) – hepatic encephalopathy (latent and expressed) the Route of administration and doses Inside on 1-2 bags of the granulate dissolved in 200 ml of liquid to 3 times a day in time or after a meal. A course of treatment from 2 weeks to 3 months depending on a course of the disease. Side effects Sometimes (≥1/1000, & lt, 1/100) – allergic symptoms in the form of the complicated breath (asthma attacks) and skin rashes – nausea, vomiting, a stomach ache, a meteorism, diarrhea Very seldom (≥1/10) – extremity pain the Specified side effects usually are passing and do not demand drug withdrawal. Contraindications – hypersensitivity to L – ornithine – L – to aspartate or other components of drug – the profound renal failure (clearance of creatine more than 3 mg / 100мл) – children’s and teenage age up to 18 years Medicinal interactions are not described Special indications of the Granule Gepa-Mertz contain fructose. Drug is not recommended to patients with rare hereditary intolerance of fructose. 1 bag of granules Gepa-Mertz contains 1.13 g of fructose (0.11 CEU is equivalent) that should be considered to the patients having diabetes. At prolonged use of a granule Gepa-Mertz can do harm to teeth (caries). Pregnancy and lactation It is necessary to avoid reception of granules Gepa-Mertz during breastfeeding and during pregnancy. Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms. Depending on a course of the disease the ability to run the vehicle or potentially dangerous mechanisms can worsen. Overdose Symptoms: strengthening of expressiveness of side effects. Treatment: gastric lavage, intake of activated carbon, symptomatic therapy. A form of release and packing On 5.0 g in bags from a multilayered (paper-polyethylene-aluminum-polyethylene) foil. On 30 bags together with the instruction for medical use in the state and Russian languages place in a pack cardboard. To Store storage conditions at a temperature not above 25 °C. To store out of children’s reach! Not to apply a period of storage of 5 years after an expiration date. Prescription status Without prescription of Proizvoditel Mertz Pharm GmbH and To.KGaA Frankfurt am Main, Germany Ph. +49-69-1503-0, fax: +49-69-1503-200, e-mail: info@merz.de the Owner of the registration certificate of Mertz Pharmasyyutikals GmbH, Frankfurt am Main, Germany the Name, the address and a contact information (phone, the fax, e-mail) of the organization in the territory of the Republic of Kazakhstan, the accepting claim (offer) on quality of medicines from consumers and responsible for post-registration observation of safety of medicine: Representative office H. Abbe Pharm GmbH (H.Abbe Pharma GmbH) in the city of Almaty of the Republic of Kazakhstan. 050002, Almaty, Zhibek Zholy St. 64/47, office 618A ph. 8 (727) 273 86 40 / 8 (727) 273 84 36 E-mail: info@abbe-pharma.kz
Additional information
| Ingredient |
|---|



















Reviews
There are no reviews yet.