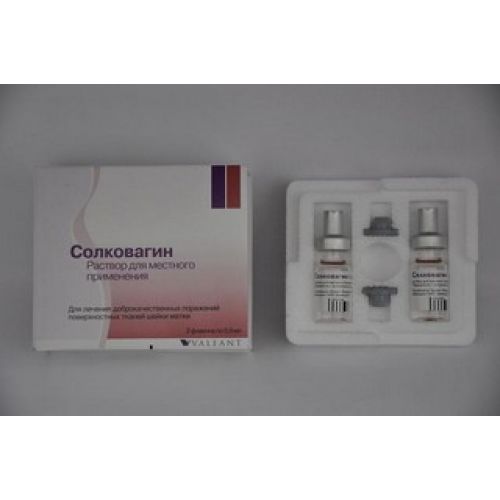
Solcovagyn 0.5 ml, 2’s solution (external application)
$53.40
Out of stock
Description
The instruction for use
of medicine for experts
of SOLKOVAGIN
the Trade name
Solkovagin
Mezhdunarodnoye the unlicensed name
Is not present
the Dosage form
Solution for topical administration
Structure
1 ml of solution contains
active agents: acetic acid of 99% 20.4 mg
oxalic acid dihydrate of 58.6 mg
nitrate zinc hexahydrate of 6.0 mg
nitric acid of 70% 537.0 mg,
excipients: purified water.
Description
Transparent colourless solution.
Pharmacotherapeutic group
Other drugs for treatment of gynecologic diseases.
Code of automatic telephone exchange G02CX
Pharmacological
Drug of Local Action properties. The composition of the drug Solkovagin developed especially for treatment of benign defeats of surface tissues of neck of the uterus causes unequal impact of drug on various types of an epithelium of a neck of the uterus. At applique nekrotizirut a malrelated cylindrical epithelium and a subepithelial stroma of an erosion. Subsequently nekrotizirovanny fabric is replaced with a multilayered flat epithelium.
Indications
Benign damages of an epithelium of a neck of the uterus
– a polyp of a neck of the uterus
– an erosion and ectropion of a neck of the uterus
the Route of administration and doses
Drug is intended only for topical administration in the field of surface tissues of a neck of the uterus. The procedure has to be carried out only by the expert – the gynecologist (using a kolposkop). During the procedure it is necessary to process only a defeat zone, avoiding hit of solution on skin of external genitals and an epithelium of a vagina.
1. To remove vaginal slime with a cotton plug. Processing of the struck zone of 3% solution of acetic acid gives the chance of more accurate visual delimitation of defeat.
2. To impregnate with Solkovagin’s solution the small cotton plug wound on a wooden stick. To process a defeat zone.
3. In 1-2 minutes to repeat processing by means of the fresh cotton plug impregnated with Solkovagin’s solution. As shows experience, such double processing enhances therapeutic effect.
4. Treatment by Solkovagin is carried out on an outpatient basis and does not provide any restrictions in acceptance of hydrotherapeutic procedures and maintaining sex life.
5. Control surveys are made according to the scheme: 1 survey – in 10 days after holding a procedure, the 2nd survey – in 14 days after carrying out 1 survey, the 3rd survey – in 14 days after carrying out 2 surveys. In case of unsatisfactory result of inspection, it is necessary to carry out a repeated course of treatment (2 procedures) and to make the next control surveys according to the above-stated scheme.
By-effects
– local reaction (irritation)
of the Contraindication
– malignant changes of a neck of the uterus
– a cellular dysplasia
– pregnancy
– hypersensitivity to drug components
Medicinal interactions
Solkovagin’s Interaction with other medicines of local action is not established.
Special instructions
of Solkovagin is the drug containing concentrated acids. It is necessary to avoid hit of drug on clothes, skin and especially – in eyes. In case of hit of Solkovagin solution on skin of external genitals or an epithelium of a vagina it is necessary to wash a drug hit zone with water immediately. At hit of Solkovagin solution on integuments or in eyes it is necessary to wash immediately a drug hit zone a plentiful amount of water and if it is possible, with neutralized solution, such as 1% Natrii hydrocarbonas solution.
Use in pediatrics
Drug is not intended for use in pediatrics.
Order of use and destruction of packing.
1. To open a bottle cover.
2. To lower the cotton plug which is reeled up on a wooden core in a bottle. Contents of one bottle are counted on holding two procedures.
3. After holding a procedure to place the used cotton plug in a bottle approximately on 1 cm and to break off a core.
4. To close a bottle the rubber stopper enclosed in packing in order to avoid hit of residues of solution on surrounding objects, then to throw out in a trash bin.
Influence on ability to run the vehicle or potentially dangerous mechanisms
does not influence.
Overdose
Symptoms: at excess use of drug the developing of burns is possible that can lead to pathological changes of an epithelium of a neck of the uterus.
Treatment is symptomatic.
A form of release and packing
On 0.5 ml of solution in the bottles of colourless glass with a nominal volume of 2 ml corked by rubber bungs with a teflon covering, with a running in caps aluminum.
2 bottles complete with two additional rubber bungs are located in the container from polyfoam containing nests for the corked bottles and a nest for the opened bottle.
A container together with the instruction for use are located in a cardboard pack.
To Store storage conditions at a temperature from + 150C to + 25 0C.
Attention! Solution possesses the strong corroding action. To handle care!
To store out of children’s reach!
Formation of a crystal deposit is possible that is a consequence of storage of drug at lower temperatures, for example, during transportation. Short-term heating of drug (1-2 min.) at 400C leads to dissolution of a deposit.
3 years
not to use a period of storage after the termination of the period of storage specified on packing.
Prescription status
According to the prescription
the Producer
of Valeant Pharmasyyutikals Switzerland GmbH,
Switzerland Rurbergstrasse 21, SN-4127 Birsfelden (Switzerland)


















Reviews
There are no reviews yet.