Description
The instruction for medical use
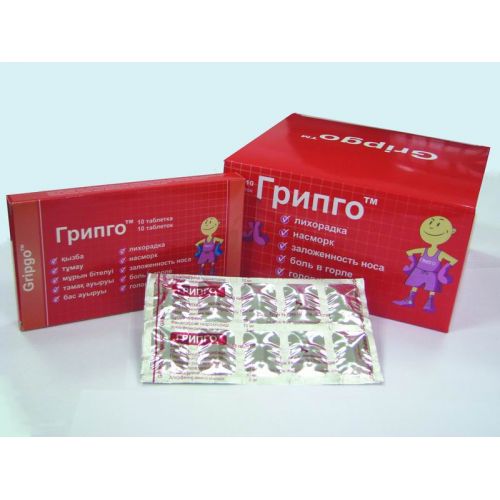
of GRIPGO medicine
A trade name
of GRIPGO
the International unlicensed name
Is not present
the Dosage form
of the Tablet Structure One Tablet contains
active agents: paracetamol of granules of 96% (it is equivalent to paracetamol of 500 mg), caffeine of anhydrous 30 mg, Phenylephrinum a hydrochloride of 10 mg, chlorpheniramine a maleate of 2 mg
excipients: microcrystalline cellulose, sodium of a kroskarmelloz, magnesium stearate.
Description
Biconvex tablets of a capsuloobrazny form of white color. Marbling is allowed.
Pharmacotherapeutic group
Other analgetics and antipyretics. Anilides. Paracetamol in a combination with other drugs excepting psikholeptik.
The ATX N02BE51 code
the Pharmacological
Pharmacokinetics Paracetamol properties is quickly and almost completely absorbed from digestive tract. The maximum concentration it in blood plasma is reached in 2 hours after oral administration. About 25% of paracetamol contact proteins of blood plasma. Paracetamol is metabolized in a liver and removed mainly with urine in the form of glyukuronidny and sulphatic conjugates. In not changed look less than 5% are removed. Elimination half-life makes 1 4 hour.
Caffeine is quickly absorbed and distributed on all bodies and body tissues, including central nervous system, embryonic fabrics and breast milk. It is quickly metabolized and removed mainly with urine (70%).
Phenylephrinum is quickly soaked up after oral administration, peak concentration in blood plasma are reached in 1 2 hour, the level of absorption of 70%, elimination half-life of 2 3 hour, is removed mainly with urine.
Chlorpheniramine the maleate is quickly absorbed from digestive tract and distributed on all body tissues. About 25% contact plasma proteins.
Gripgo’s pharmacodynamics is the combined medicine which effect is caused by the components which are its part.
Paracetamol is the analgetic-antipyretic having the febrifugal and anesthetizing properties and also the insignificant anti-inflammatory effect connected with influence of paracetamol on the hypothalamic center of thermal control and its opportunity to inhibit synthesis of prostaglandins.
Caffeine is an alkaloid of group of methylxanthines which directly stimulates the respiratory and vasomotor centers of a brain, improves a physical and emotional condition of the patient thanks to what manifestations of an asthenia in infectious diseases decrease. Increases analgesic effect of paracetamol.
Phenylephrinum a hydrochloride adrenomimetichesky means which reduces hypostasis and hyperaemia in upper parts of airways and sine that promotes improvement of nasal breath. Stimulates mainly alpha adrenoceptors thanks to what there is a narrowing of peripheral vessels and reduction of their permeability, formation of a mucous secret decreases.
Chlorpheniramine a maleate antihistamine which reduces an allergic component of an infectious disease. Competitively blocks histamine H1 receptors and interferes with development of effects of a histamine, eliminates cold, an itching of a nose and gripes in eyes.
Indications
– the treatment of symptoms of flu and other acute respiratory viral diseases which are followed by fever, head and muscular pain, cold, an allergic component in the form of congestion of a nose, sneezing, dacryagogue
the Route of administration and doses
to Adults and children are more senior than 12 years appoint on 1 tablet 3 4 of time a day, to wash down with water. Maximum daily dose 4 tablets. Duration of a course of treatment is no more than 3 5 day. It is not recommended to exceed the specified doses.
Side effects
– skin rash, an itching, a small tortoiseshell, a Quincke’s disease
– dizziness, backfilling disturbance, hyperexcitability
– increase in arterial blood pressure, tachycardia
– nausea, vomiting, epigastric pain, dryness in a mouth
– an ischuria
– accommodation paresis, increase in intraocular pressure, a mydriasis
– anemia, thrombocytopenia, an agranulocytosis, hemolytic anemia, aplastic anemia, a methemoglobinemia, a pancytopenia
– hepatotoxic action
– nephrotoxicity (papillary necrosis)
– a bronkhoobstruktion.
– at prolonged use, especially in high doses, development of a methemoglobinemia, anemia, nephrotoxic and hepatotoxic action is possible.
With care – the profound atherosclerosis of coronary arteries, arterial hypertension, a thyrotoxicosis, a pheochromocytoma, diabetes, bronchial asthma, a chronic obstructive pulmonary disease,
deficit glucose – 6 fosfatdegidrogenaza, blood diseases, congenital hyperbilirubinemias (Gilbert’s syndromes, the Cudgel Johnson and the Rotor), a liver and/or renal failure, closed-angle glaucoma, a prostate hyperplasia.
Contraindications
– hypersensitivity to drug components
– a concomitant use of tricyclic antidepressants, MAO inhibitors, beta blockers
– a concomitant use of the drugs containing paracetamol
– arterial hypertension
– a hyperthyroidism
– a prostatauxe
– an alcoholism
– a disease of kidneys and a liver
– pregnancy and the period of a lactation
– children’s age up to 12 years
Medicinal interactions
it is not necessary to appoint Gripgo with other drugs containing paracetamol and also other non-steroidal anti-inflammatory drugs.
Risk of development of hepatotoxic effect increases at simultaneous use of barbiturates, dipheninum, carbamazepine, rifampicin, a zidovudine and other inductors of microsomal enzymes of a liver. Barbiturates reduce zharoponizhiyushchy effect of paracetamol.
It is not necessary to use at the same time drug with tricyclic antidepressants, MAO inhibitors and within 14 days after their cancellation because of a possibility of strengthening of hepatotoxic effect of paracetamol. Against the background of reserpine the increase in arterial blood pressure is possible. Alkaloids of an ergot strengthen an aritmogennost.
At a concomitant use of Metoclopramidum, a domperidon or a holestiramin the absorbability of paracetamol decreases.
Flukonazol, ketokonazol, meksiletin and terbinafin can moderately increase concentration of caffeine in plasma with possible strengthening of its effect and toxicity.
Drug enhances effect of indirect anticoagulants.
Special instructions
With care to apply at tendency of the patient to bradycardia, in heart failure, coronary heart disease, atherosclerosis, arterial hypertension, a prostatauxe, to people of advanced age.
Control of blood tests is necessary for patients with pathology of a system of blood. During the Gripgo drug treatment the consumption of alcoholic beverages is strictly forbidden.
The feature of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
during Gripgo’s reception should be careful at control of vehicles and work with potentially dangerous mechanisms that is caused by a possibility of reduction in the rate of reactions.
Overdose
Symptoms: nausea, vomiting, dryness in a mouth, a nose and a throat, pain in a stomach or a liver, perspiration, breath disturbance, confusion of consciousness, bradycardia, arterial hypertension, tachycardia, arrhythmia, face reddening. In hard cases the liver failure and a coma develops.
Treatment: gastric lavage and purpose of sorbents, symptomatic therapy is in case of need carried out. Antidote of paracetamol is Acetylcysteinum, methionine.
To Store storage conditions in the dry, protected from light place, at a temperature not over 25C.
To store out of children’s reach!
A period of storage
3 years
Prescription status
Without prescription
the Form of release and packing
On 4 tablets in planimetric bezjyacheykovy packing from the printing aluminum foil varnished or in blister strip packaging from transparent PVH/PVDH of a film and aluminum foil.
On 1 planimetric bezjyacheykovy or blister strip packaging together with the instruction for use in the state and Russian languages place in a paper envelope.
On 50 envelopes place in a box of cardboard.
On 10 tablets in planimetric bezjyacheykovy packing from the printing aluminum foil varnished or in blister strip packaging from transparent PVH/PVDH of a film and aluminum foil.
On 1 planimetric bezjyacheykovy or blister strip packaging together with the instruction for use in the state and Russian languages place in individual cardboard packing.
On 10 individual cardboard packs place in a box of cardboard.
On 10 planimetric bezyacheykovy or blister strip packagings together with the instruction for use in the state and Russian languages place in a box of cardboard.
Producer Kusum Heltker Pvt.
Ltd., JV 289 (A), Riiko Indl. Chopanki’s Area, Bkhivadi (Raj.), India
Owner of the registration certificate
Kusum Heltker Pvt. Ltd., India
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products:
Representative office of Kusum Heltker Pvt Association. Ltd., India in the Republic of Kazakhstan,
legal address: Almaty, Dostyk Ave., 117/6, BC Khan Tengri
the location address: Almaty, Dostyk Ave., 117/6, BC Khan Tengri.
Phone number: 295-26-50, 295-26-51 (54), Fax: 295-26-55
E-mail:
To develop Office@kusumhealthcare.kz


















Reviews
There are no reviews yet.