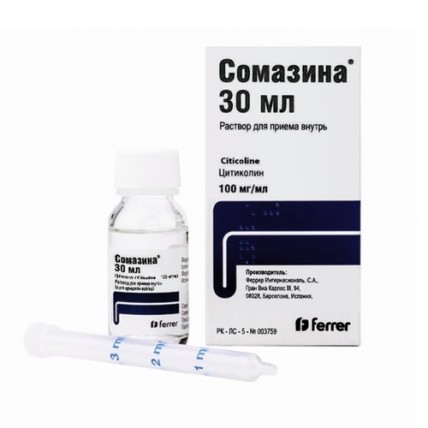
Somazina® (Citicoline) 100 mg/ml, 30 ml oral solution
- $46.00
What is Somazina® (Citicoline) 100 mg/ml?
Somazina® is an oral solution containing Citicoline, an active compound that supports brain health and function.
Citicoline is known for its neuroprotective and nootropic effects, which means it can help improve cognitive function and protect the brain from damage caused by various factors.
This medication is commonly used to support recovery from brain injuries, improve cognitive deficits, and assist in the rehabilitation of patients who have suffered from cerebrovascular accidents, such as strokes.
How does Somazina® work?
Citicoline, the active ingredient in Somazina®, works by increasing the levels of important chemicals in the brain that help repair and protect brain cells.
It enhances the synthesis of phospholipids, which are essential components of the cell membranes, and helps restore the integrity of damaged neuronal membranes.
This action supports brain health, especially after injuries or in conditions that cause cognitive decline.
What are the common uses of Somazina®?
Somazina® is used in a variety of situations where brain function needs support.
The main indications include:
- Acute and recovery phases of cerebrovascular accidents: It aids in the rehabilitation process following strokes by supporting cognitive and motor function recovery.
- Traumatic brain injuries and their sequelae: It helps manage the symptoms and promote recovery after head injuries.
- Cognitive, sensory, motor, and neuropsychological disorders: These conditions, often resulting from degenerative or vascular issues in the brain, can benefit from the neuroprotective effects of Citicoline.
Who should avoid taking Somazina®?
Certain individuals should not use Somazina®, including those who:
- Have allergies to any component of the medication.
- Suffer from conditions related to the parasympathetic nervous system.
- Have inherited fructose intolerance or other related metabolic disorders.
- Are children or adolescents under 18 years of age due to the presence of the dye Ponceau 4R (E124), which is not recommended for this age group.
Are there any special precautions for using Somazina®?
While Somazina® is generally safe, there are some considerations to keep in mind:
- Allergic Reactions: Some individuals may experience allergic reactions due to the presence of Ponceau 4R, methylparaben, or propylparaben in the formulation.
- Fructose Intolerance: The solution contains sorbitol, so it should not be used by individuals with hereditary fructose intolerance.
- Asthma: Patients with a history of asthma or aspirin allergy may be at increased risk of developing bronchial asthma when using this product.
Can Somazina® be used during pregnancy or breastfeeding?
There is limited data on the use of Citicoline during pregnancy or breastfeeding.
Therefore, it should only be used if the potential benefits justify the potential risks to the mother and baby.
Consultation with a healthcare provider is essential before using this medication during pregnancy or lactation.
How should Somazina® be taken?
Somazina® is an oral solution that can be taken either directly or diluted in half a glass of water (approximately 120 ml). The dosage varies depending on the severity of the condition but typically ranges from 500 mg to 2000 mg per day. The medication can be taken with or without food, and it is administered using a dosing syringe, which should be rinsed with water after each use.
What should you do if you miss a dose?
If you miss a dose of Somazina®, take it as soon as you remember.
However, if it is almost time for your next dose, skip the missed dose and continue with your regular schedule.
Do not double the dose to make up for the missed one.
What are the possible side effects of Somazina®?
While side effects are rare, some individuals may experience:
- Headache or dizziness
- Blood pressure changes (either high or low)
- Respiratory issues such as shortness of breath
- Gastrointestinal symptoms like nausea, vomiting, or diarrhea
- Skin reactions including rash, hives, or redness
If you experience any of these symptoms, consult your healthcare provider for advice.
How should Somazina® be stored?
Store Somazina® at a temperature not exceeding 30°C (86°F).
Do not freeze or refrigerate the solution.
Always keep it out of reach of children and do not use it after the expiration date printed on the package.