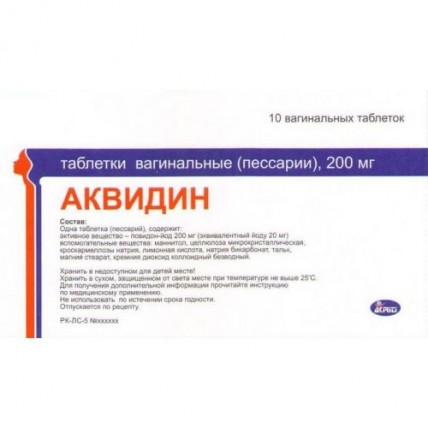
Akvidin 200 mg vaginal (10 tablets)
- $14.10
Sku:
7fb14f106f8c
The instruction for medical use of Akvidin Torgovoye medicine a name Akvidin Mezhdunarodnoye the unlicensed name Is not present the Dosage form of the Tablet vaginal (pessaries), 200 mg Structure One tablet (pessary), contains active agent – povidone-iodine of 200 mg (equivalent to iodine of 20 mg) excipients: Mannitolum, cellulose microcrystalline, croscarmellose sodium, citric acid, sodium bicarbonate, talc, magnesium stearate, silicon dioxide colloidal anhydrous Description of the Tablet (pessaries) of an almond-shaped form, light brown color, biconvex, not coated. Pharmacotherapeutic group of Antiseptics and antimicrobial drugs for treatment of gynecologic diseases (including combinations with corticosteroid). Other antiseptic agents and antimicrobial drugs. Povidone – iodine. ATX G01AX11 code the Pharmacological Pharmacokinetics At properties contact with a mucous membrane of a vagina iodine is gradually and evenly released (only 30% of a complex turn into iodides). At iodine absorption the volume of distribution is 38%, elimination half-life makes 2 days. It is allocated with kidneys. With normal function of a thyroid gland the increased absorption of iodine does not cause any significant changes of its hormonal function in patients. Iodine gets through a placental barrier and is emitted in breast milk. Absorption of povidone and its discharge depends on molecular weight the Pharmacodynamics Drug renders antiseptic, disinfecting, antifungal and anti-protozoan action. Possesses a wide range of antimicrobial action. It is active concerning Gram-positive and Gram-negative bacteria, Gardnerella vaginalis, mycoplasma, Treponema pallidum, chlamydias, mushrooms (for example, the sorts Candida), viruses, the elementary (including trichomonads) and sporoshok Active agent - iodine - is provided in the form of a PVP-iodine complex. From this complex elementary iodine is gradually and evenly released that provides constant concentration of active free iodine. Concentration of active iodine makes 0.1-1%. Action mechanism: at contact with proteins of a bacterial wall or fermental proteins forms iodamines and, blocking their action, has bactericidal effect on microorganisms. Drug facilitates restoration of normal flora of a vagina. Due to gradual release of iodine, drug works is long. In a complex with polyvinylpirrolidone iodine considerably loses local irritative action and therefore it is well transferred when drawing to mucous membranes, skin and wound surfaces. Indications Acute and persistent vaginal infections, multi-infections: - nonspecific infections (bacterial vaginosis, Gardnerella vaginalis), fungal infections (Candida albicans), including candidiasis owing to treatment by antibiotics and steroid drugs - genital herpes - trichomoniasis (if necessary it is necessary to carry out the combined system treatment). - Preoperative and postoperative processing (for the purpose of prevention) before surgeries on a vagina and also obstetric and diagnostic procedures. A route of administration and doses For intravaginalny introduction. Adult and elderly people After preliminary hygienic processing to enter on 1 tablet is deep in a vagina, previously having moistened a tablet with water. In an acute vaginitis 1-2 times a day within 7 days, in a chronic and subacute vaginitis - 1 time a day before going to bed within 14 days (it is possible longer). Throughout a course of treatment it is recommended to use sanitary pads. It is not necessary to stop use of tablets (pessary) during periods. Children: It is contraindicated for use at children of prepubertatny age. Side effects - hypersensitivity reactions to drug - hyperaemia - an itching. In rare instances it can cause: - reactions of hypersensitivity in the form of contact dermatitis with formation of psoriazopodobny red small bullous elements - the hyperthyroidism at predisposed persons induced by iodine at prolonged use of vaginal suppositories. Contraindications - hypersensitivity to iodine and other components of drug - dysfunction of a thyroid gland (the nodal colloidal craw, an endemic craw also tiroidit Hashimoto, a hyperthyroidism) - adenoma of a thyroid gland, herpetiform dermatitis of Dyuringa - simultaneous use of radioiodine - the children's age before puberty Medicinal interactions is not compatible to another disinfecting and the antiseptics which are especially containing alkalis, enzymes and mercury. Combined use povidone of iodine and peroxide of hydrogen and also the fermental drugs containing silver and tauloridin for processing of wounds and also antiseptic drugs, leads to mutual decrease in efficiency. In the presence of blood the bactericidal action can decrease, however at increase in concentration of solution, the bactericidal activity can be increased. Special instructions Due to the oxidizing properties can result povidone-iodine, its traces in false positive results of some types of researches for detection of the occult blood in Calais and also blood or glucose in urine. During use, povidone-iodine can decrease iodine absorption by a thyroid gland that can affect results of some diagnostic tests (for example, stsintigrafiya of a thyroid gland, definition of the proteinaceous and connected iodine, researches using radioiodine) and also can interact with the iodine drugs used for treatment of diseases of a thyroid gland. For obtaining not distorted results of a stsintigrafiya of a thyroid gland after long therapy povidone-iodine is recommended to sustain rather long span without use of this drug. If during a course of treatment there are hyperthyroidism symptoms, it is necessary to check function of a thyroid gland. It is necessary to be careful at regular use of drug for patients with earlier diagnosed renal failure. It is necessary to avoid regular use of vaginal tablets (pessaries) Akvidin at the patients receiving lithium drugs. Coloring on skin and fabrics easily washes off. After contact with drug it is necessary to avoid its hit in eyes. Tablets possess spermicidal action, in communication with what their use for the persons planning pregnancy is not recommended. During use of tablets it is possible to recommend use of sanitary pads. Pregnancy and feeding by a breast Iodine gets through a placental barrier and is allocated in breast milk. Use of drug during pregnancy can cause development of a tranzitorny hypothyroidism in a fruit and the newborn. Use of drug at duration of gestation over two months and also during breastfeeding is possible only according to strict indications and demands careful observation of function of a thyroid gland of mother and the child. In these cases it is possible to use drug only quickly. Use of drug from the second trimester of pregnancy is not recommended. Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms Use of drug does not affect ability to driving of motor transport and control of potentially dangerous mechanisms. Overdose the following clinical manifestations are characteristic Of acute iodic intoxication. Symptoms: the metal taste in a mouth, the increased salivation, burning sensation or mouth or throat pain, irritation also swelled an eye, skin reactions, gastrointestinal disorders and a diarrhea, a renal failure and an anury, a circulatory unefficiency, a laryngeal edema with secondary asphyxia, a fluid lungs, a metabolic acidosis, a hypernatremia. Treatment: drug withdrawal, symptomatic and maintenance therapy with special control of electrolytic balance, function of kidneys and a thyroid gland. Toxic concentration of iodine in blood serum can be reduced effectively peritoneal dialysis or a hemodialysis. Besides, careful clinical observation of function of a thyroid gland has to be established for an exception or early detection of the hyperthyroidism caused by iodine. Intoxication after intake: immediate introduction of the foodstuff containing starch or protein (for example, starch solution in water or milk). Gastric lavage by 5% solution of sodium thiosulphate (or intravenous administration of 10 ml of 10% solution of sodium thiosulphate) with three-hour intervals as required. A form of release and packing On 10 tablets (pessaries) in blister strip packagings from aluminum foil (Alu | Alu). On 1 blister strip packaging complete with the applicator and the instruction for use in the state and Russian languages put in a pack cardboard. To Store storage conditions in the dry, protected from light place at a temperature not higher than 25 wasps. To store out of children's reach! 3 years not to use a period of storage after an expiration date. Prescription status According to the prescription the Producer Akriti Pharmasyyutikalz Pvt. Ltd., India Plot No. D-10, D-11, M.I.D.C. Jejury, Dist. Pune - 412,303, Maharashtra the Name and the country of the owner of the registration certificate of Avitsena-Ltd LLP, Kazakhstan the Name and the country of the Akriti Pharmasyyutikalz Pvt organization packer. Ltd., India and
to Develop the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products of Avitsena-LTD LLP, 50002, Almaty, Dzhangildin St., 31, office 416 the Address of the organization responsible for post-registration observation of safety of medicine of ConsultAsia LLP, Shevchenko St. of 165 B, office 909
to Develop the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products of Avitsena-LTD LLP, 50002, Almaty, Dzhangildin St., 31, office 416 the Address of the organization responsible for post-registration observation of safety of medicine of ConsultAsia LLP, Shevchenko St. of 165 B, office 909