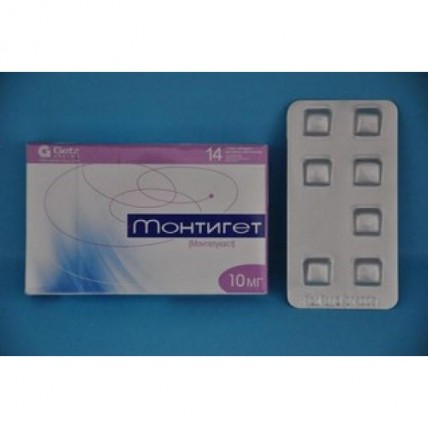
Montiget 14s 10 mg film-coated tablets
- $26.20
The instruction for medical use
of MONTIGET medicine
A trade name
of Montiget Mezhdunarodnoye the unlicensed
name Montelukast Dosage Form
of the Tablet, film coated 10 mg
Structure
One coated tablet contains
active agent - sodium montelukast of 10.40 mg
(it is equivalent to montelukast) 10.00 mg
excipients: cellulose microcrystalline (avitset PH 102), lactose regulyar, sodium of a kroskarmelloz, a hydroksipropilmetiltsellyuloz (Pharmakoakt 606), magnesium stearate
structure of a film cover: the talc purified, the water purified, Opadri II dye yellow: the polyvinyl alcohol hydrolyzed, the talc purified the titan dioxide (E171), a macrogoal, lecithin, ferrous oxide yellow (E172), ferrous oxide black (E172).
The description
of the Tablet of light yellow color, film coated, square shape, with risky on one party and smooth on other party.
Pharmacotherapeutic group
Drugs for treatment of diseases of a respiratory system. Other drugs for treatment of obstructive respiratory diseases for system use. Leukotriene receptors antagonists. Montelukast.
ATX R03DC03 code
the Pharmacological
Pharmacokinetics Montelukast properties of sodium is quickly soaked up after intake.
The maximum plasma concentration are reached in 2-4 hours after reception. The bioavailability is 64%. More than for 99% montelukast of sodium contacts proteins of blood plasma. Plasma elimination half-life of blood makes from 3 to 5.5 hours at healthy adults. The pharmacokinetics of montelukast shows linear character up to a dose of 50 mg.
Montelukast of sodium is actively metabolized in a liver through the system of P450 cytochrome by CYP3A4, CYP2A6 and CYP2C9 isoenzymes. Therapeutic plasma concentration of montelukast of sodium do not inhibit enzymes 3A4, 2C9, 1A2, 2A6, 2C19 or 2D6.
The plasma clearance of montelukast is about 45 ml/min. at healthy adults. Montelukast of sodium is removed with excrements and bile.
Children, patients of advanced age and patients with a renal failure have similar pharmacokinetic parameters with healthy adults.
At patients with a moderate liver failure and clinical manifestations of cirrhosis the metabolism of montelukast slows down and extended elimination half-life: AUC (the area under a curve of plasma concentration) increases by 41% after reception of a dose of 10 mg.
Correction of a dosage with a renal failure is not required from patients.
Montiget's pharmacodynamics the selection antagonist of CysLT1-receptors of tsisteinilovy leukotrienes (LT C4, D4 and E4) - the most powerful mediators of the chronic persistent inflammation maintaining hyperreactivity of bronchial tubes in bronchial asthma. Bronkhodilatiruyushchy effect of montelukast is connected with blocking leukotriene D4 receptors in airways and reduction of effect of leukotrienes - the endogenous biologically active agents playing an important role in processes of regulation of reactivity of bronchial tubes, development of a bronkhokonstriktion, hypostasis of a mucous membrane of bronchial tubes, bronchial hypersecretion.
The therapeutic effect of Montiget begins to be shown during the first day of administration of drug.
Thus, montelukast blocks effect of LTD4 leukotrienes at the TsisLT1-retseptorov level without agonistic action.
Indications
- prevention and long-term treatment of bronchial asthma, including prevention of day and night symptoms of a disease, treatment of aspirinchuvstvitelny patients with bronchial asthma and prevention of the bronchospasm caused by physical activity
- treatment of day and night symptoms of seasonal allergic rhinitis
the Route of administration and
Montiget's doses is accepted once a day. The drug can be taken in time, to or after meal.
For treatment of bronchial asthma the dose of drug should be accepted in the evening.
The patients having at the same time asthma and seasonal allergic rhinitis take the drug once in the evening.
Patients should report that they should take the drug Montiget even if will reach control of asthma and also during the periods of deterioration in a course of asthma.
Adults and teenagers of 15 years are also more senior: accept 10 mg a day once a day (evening).
For elderly patients, patients with a renal failure and also to patients with slight or medium-weight abnormal liver functions and also depending on a floor of special selection of a dose it is not required.
Co-administration of Montiget in complex treatment of asthma
of Montiget can be added to treatment of the patients accepting bronchodilators and inhalation glucocorticosteroids (See the Section Interaction with other medicines).
To Montiget it is not recommended as monotherapy for patients with persistent asthma of moderate severity. Montelukast use as an alternative to low-dose inhalation corticosteroids for children with persistent asthma of light severity should be considered only for patients who had no serious attacks of asthma demanding use of oral corticosteroids and who are not capable to use inhalation corticosteroids lately. Persistent asthma of light severity is defined as emergence of symptoms of asthma more often than once a week, but less than once a day, emergence of night symptoms more often than 2 times a month, but less than once a week, normal function of lungs in the periods between episodes. If to control visit of sufficient control of asthma it is not reached (usually for 1 month), it is necessary to define need for additional or other anti-inflammatory therapy, based on the consecutive system of treatment of asthma. It is necessary to estimate periodically a condition of patients concerning control of asthma.
Montiget is appointed to patients with a bronchospasm if the clinical effect of the isolated use of bronchodilators is insufficient.
In case of achievement of adequate clinical effect (usually after reception of the first dose) the dose of the applied bronchodilators can be reduced.
Corticosteroid inhalers
If use the drug Montiget as additional therapy at treatment by inhalation corticosteroids, it is not necessary to replace with the drug Montiget inhalation corticosteroids sharply.
Side effects
Often
- nausea, vomiting, thirst, dyspepsia, diarrhea, an abdominal pain
- a headache
Seldom
- reactions of hypersensitivity: an anaphylaxis, a Quincke's disease, a small tortoiseshell, rash, an itching
- drowsiness, an insomnia, pathological dreams, hallucinations, insomnia, disturbance of attention, a memory impairment
- psychomotor hyperactivity (irritability, excitement, agressive behavior)
- an asthenia, fatigue, weakness, an indisposition, hypostases
- arthralgias, myalgias, spasms of muscles
- bleeding, formation of hypodermic hemorrhages
- eosinophilic infiltrates of a liver
- the increased content of transaminases in blood serum
(alaninaminotranspherases, aspartate aminotransferases)
is Very rare
- cholestatic hepatitis
- spasms
- a disorientation, suicide thoughts and behavior (suitsidalnost)
- Cherdzha-Stross
Protivopokazaniya's syndrome
- the known hypersensitivity to montelukast of sodium or other components of drug
- pregnancy and the period of a lactation
- children up to 15 years
- hereditary insufficiency of lactose and also intolerance of sucrose, fructose and galactose
it is possible to appoint Medicinal interactions of Montiget together with other medicines which are traditionally applied to prevention and long-term treatment of bronchial asthma.
Sharp replacement of therapy with inhalation corticosteroids with prescribing of montelukast is not recommended.
The recommended clinical dose of montelukast does not render clinically significant effect on pharmacokinetics of the following drugs: theophylline, Prednisolonum, oral contraceptives (ethinylestradiol/norethindrone 35/1), terfenadina, digoxin and warfarin.
AUC decreases at the persons who are at the same time receiving phenobarbital (approximately by 40%), however correction of the mode of dosing Montigety is not required to such patients.
As montelukast is metabolized by means of CYP 3A4, it is necessary to appoint with care drug, especially to children, along with inductors CYP 3A4, such as Phenytoinum, phenobarbital and rifampicin.
Special instructions
It is necessary to observe the treatment mode accurately.
Montelukast should not be used for stopping of bad asthmatic attacks (does not replace inhalation bronchodilators), at emergence of therapeutic effect (usually after the first dose) the number of inhalations of bronchodilators can be within a day reduced.
It is impossible to replace with montelukast inhalation or oral corticosteroids sharply. However the dose of an inhalation corticosteroid can be reduced gradually under medical observation.
Montelukast should not be used as monotherapy for treatment and control of asthma which is shown at physical activity. Patients who have strengthening of asthma after physical activity have to continue use of their usual mode of inhalation - agonists for prevention and have to have an inhaler at themselves when it is required.
Seldom the patients receiving treatment by antiasthmatic drugs, including montelukast, can have a system eosinophilia, sometimes with clinical manifestations of a vasculitis characteristic of Cherdzha-Stross's syndrome (state at which treatment is often carried out by system corticosteroids). These cases, as a rule, but not always, are connected with decrease or cancellation of oral corticosteroids. Probability that antagonists of receptors of leukotriene can be connected with emergence of a syndrome of Cherdzha-Stross cannot be excluded or confirmed. Doctors have to remember possibility at patients of an eosinophilia, vascular rash, deterioration in pulmonary symptoms, cardiological complications and/or neuropathy. Patients who had these symptoms it is necessary to survey and revise their scheme of treatment repeatedly.
Montelukast does not block the bronkhospastichesky answer to aspirin or to NPVS at the asthmatic patients sensitive to aspirin. Such patients have to continue to avoid intake of aspirin and various NPVS. The caution has to be observed when using montelukast together with bronchodilatory therapy. When the clinical answer is obvious, bronchodilatory therapy has to be reduced.
Use in pediatric practice
This dosage form is not appointed to children up to 15 years
of Feature of influence of medicine on ability to run the vehicle or potentially dangerous
Montiget mechanisms the car does not affect ability of the patient of a message or to operate various mechanisms. However, seldom or never, reported about drowsiness emergence therefore it is necessary to be careful when performing these types of activity.
Overdose
Symptoms: the abdominal pain, drowsiness, thirst, a headache, vomiting or psychomotor hyperactivity are possible.
Treatment: symptomatic. There are no data on a possibility of removal of montelukast by peritoneal dialysis or a hemodialysis.
A form of release and packing
On 7 tablets in blister strip packaging from aluminum foil. On the 2nd blister strip packagings of the blister together with the instruction for medical use in the state and Russian languages place in a cardboard box.
To Store storage conditions in the dry and protected from light place, at
a temperature from 15 of 0C up to 30 0C.
To store in the inaccessible place for children!
3 years
not to apply a period of storage on expiry date.
Prescription status
According to the prescription
the Producer Getz of Pharm (Pvt.) Limited, Pakistan,
29-30/27, K. I.A., Karachi 74900.
Ph. + (92-21) 111,111,511
Fax: + (92-21) 350 5792
www.getzpharma.com
Owner of the registration certificate
Getz of Pharm (Pvt.) Limited, Pakistan.
The address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products:
Representative office Getz of Pharm (Pvt.) Limited in
Almaty, Shevchenko St., 148 RK.
ph. +7 (727) 378-51-89, 378-54-78
www.getzpharma.com
To develop
of MONTIGET medicine
A trade name
of Montiget Mezhdunarodnoye the unlicensed
name Montelukast Dosage Form
of the Tablet, film coated 10 mg
Structure
One coated tablet contains
active agent - sodium montelukast of 10.40 mg
(it is equivalent to montelukast) 10.00 mg
excipients: cellulose microcrystalline (avitset PH 102), lactose regulyar, sodium of a kroskarmelloz, a hydroksipropilmetiltsellyuloz (Pharmakoakt 606), magnesium stearate
structure of a film cover: the talc purified, the water purified, Opadri II dye yellow: the polyvinyl alcohol hydrolyzed, the talc purified the titan dioxide (E171), a macrogoal, lecithin, ferrous oxide yellow (E172), ferrous oxide black (E172).
The description
of the Tablet of light yellow color, film coated, square shape, with risky on one party and smooth on other party.
Pharmacotherapeutic group
Drugs for treatment of diseases of a respiratory system. Other drugs for treatment of obstructive respiratory diseases for system use. Leukotriene receptors antagonists. Montelukast.
ATX R03DC03 code
the Pharmacological
Pharmacokinetics Montelukast properties of sodium is quickly soaked up after intake.
The maximum plasma concentration are reached in 2-4 hours after reception. The bioavailability is 64%. More than for 99% montelukast of sodium contacts proteins of blood plasma. Plasma elimination half-life of blood makes from 3 to 5.5 hours at healthy adults. The pharmacokinetics of montelukast shows linear character up to a dose of 50 mg.
Montelukast of sodium is actively metabolized in a liver through the system of P450 cytochrome by CYP3A4, CYP2A6 and CYP2C9 isoenzymes. Therapeutic plasma concentration of montelukast of sodium do not inhibit enzymes 3A4, 2C9, 1A2, 2A6, 2C19 or 2D6.
The plasma clearance of montelukast is about 45 ml/min. at healthy adults. Montelukast of sodium is removed with excrements and bile.
Children, patients of advanced age and patients with a renal failure have similar pharmacokinetic parameters with healthy adults.
At patients with a moderate liver failure and clinical manifestations of cirrhosis the metabolism of montelukast slows down and extended elimination half-life: AUC (the area under a curve of plasma concentration) increases by 41% after reception of a dose of 10 mg.
Correction of a dosage with a renal failure is not required from patients.
Montiget's pharmacodynamics the selection antagonist of CysLT1-receptors of tsisteinilovy leukotrienes (LT C4, D4 and E4) - the most powerful mediators of the chronic persistent inflammation maintaining hyperreactivity of bronchial tubes in bronchial asthma. Bronkhodilatiruyushchy effect of montelukast is connected with blocking leukotriene D4 receptors in airways and reduction of effect of leukotrienes - the endogenous biologically active agents playing an important role in processes of regulation of reactivity of bronchial tubes, development of a bronkhokonstriktion, hypostasis of a mucous membrane of bronchial tubes, bronchial hypersecretion.
The therapeutic effect of Montiget begins to be shown during the first day of administration of drug.
Thus, montelukast blocks effect of LTD4 leukotrienes at the TsisLT1-retseptorov level without agonistic action.
Indications
- prevention and long-term treatment of bronchial asthma, including prevention of day and night symptoms of a disease, treatment of aspirinchuvstvitelny patients with bronchial asthma and prevention of the bronchospasm caused by physical activity
- treatment of day and night symptoms of seasonal allergic rhinitis
the Route of administration and
Montiget's doses is accepted once a day. The drug can be taken in time, to or after meal.
For treatment of bronchial asthma the dose of drug should be accepted in the evening.
The patients having at the same time asthma and seasonal allergic rhinitis take the drug once in the evening.
Patients should report that they should take the drug Montiget even if will reach control of asthma and also during the periods of deterioration in a course of asthma.
Adults and teenagers of 15 years are also more senior: accept 10 mg a day once a day (evening).
For elderly patients, patients with a renal failure and also to patients with slight or medium-weight abnormal liver functions and also depending on a floor of special selection of a dose it is not required.
Co-administration of Montiget in complex treatment of asthma
of Montiget can be added to treatment of the patients accepting bronchodilators and inhalation glucocorticosteroids (See the Section Interaction with other medicines).
To Montiget it is not recommended as monotherapy for patients with persistent asthma of moderate severity. Montelukast use as an alternative to low-dose inhalation corticosteroids for children with persistent asthma of light severity should be considered only for patients who had no serious attacks of asthma demanding use of oral corticosteroids and who are not capable to use inhalation corticosteroids lately. Persistent asthma of light severity is defined as emergence of symptoms of asthma more often than once a week, but less than once a day, emergence of night symptoms more often than 2 times a month, but less than once a week, normal function of lungs in the periods between episodes. If to control visit of sufficient control of asthma it is not reached (usually for 1 month), it is necessary to define need for additional or other anti-inflammatory therapy, based on the consecutive system of treatment of asthma. It is necessary to estimate periodically a condition of patients concerning control of asthma.
Montiget is appointed to patients with a bronchospasm if the clinical effect of the isolated use of bronchodilators is insufficient.
In case of achievement of adequate clinical effect (usually after reception of the first dose) the dose of the applied bronchodilators can be reduced.
Corticosteroid inhalers
If use the drug Montiget as additional therapy at treatment by inhalation corticosteroids, it is not necessary to replace with the drug Montiget inhalation corticosteroids sharply.
Side effects
Often
- nausea, vomiting, thirst, dyspepsia, diarrhea, an abdominal pain
- a headache
Seldom
- reactions of hypersensitivity: an anaphylaxis, a Quincke's disease, a small tortoiseshell, rash, an itching
- drowsiness, an insomnia, pathological dreams, hallucinations, insomnia, disturbance of attention, a memory impairment
- psychomotor hyperactivity (irritability, excitement, agressive behavior)
- an asthenia, fatigue, weakness, an indisposition, hypostases
- arthralgias, myalgias, spasms of muscles
- bleeding, formation of hypodermic hemorrhages
- eosinophilic infiltrates of a liver
- the increased content of transaminases in blood serum
(alaninaminotranspherases, aspartate aminotransferases)
is Very rare
- cholestatic hepatitis
- spasms
- a disorientation, suicide thoughts and behavior (suitsidalnost)
- Cherdzha-Stross
Protivopokazaniya's syndrome
- the known hypersensitivity to montelukast of sodium or other components of drug
- pregnancy and the period of a lactation
- children up to 15 years
- hereditary insufficiency of lactose and also intolerance of sucrose, fructose and galactose
it is possible to appoint Medicinal interactions of Montiget together with other medicines which are traditionally applied to prevention and long-term treatment of bronchial asthma.
Sharp replacement of therapy with inhalation corticosteroids with prescribing of montelukast is not recommended.
The recommended clinical dose of montelukast does not render clinically significant effect on pharmacokinetics of the following drugs: theophylline, Prednisolonum, oral contraceptives (ethinylestradiol/norethindrone 35/1), terfenadina, digoxin and warfarin.
AUC decreases at the persons who are at the same time receiving phenobarbital (approximately by 40%), however correction of the mode of dosing Montigety is not required to such patients.
As montelukast is metabolized by means of CYP 3A4, it is necessary to appoint with care drug, especially to children, along with inductors CYP 3A4, such as Phenytoinum, phenobarbital and rifampicin.
Special instructions
It is necessary to observe the treatment mode accurately.
Montelukast should not be used for stopping of bad asthmatic attacks (does not replace inhalation bronchodilators), at emergence of therapeutic effect (usually after the first dose) the number of inhalations of bronchodilators can be within a day reduced.
It is impossible to replace with montelukast inhalation or oral corticosteroids sharply. However the dose of an inhalation corticosteroid can be reduced gradually under medical observation.
Montelukast should not be used as monotherapy for treatment and control of asthma which is shown at physical activity. Patients who have strengthening of asthma after physical activity have to continue use of their usual mode of inhalation - agonists for prevention and have to have an inhaler at themselves when it is required.
Seldom the patients receiving treatment by antiasthmatic drugs, including montelukast, can have a system eosinophilia, sometimes with clinical manifestations of a vasculitis characteristic of Cherdzha-Stross's syndrome (state at which treatment is often carried out by system corticosteroids). These cases, as a rule, but not always, are connected with decrease or cancellation of oral corticosteroids. Probability that antagonists of receptors of leukotriene can be connected with emergence of a syndrome of Cherdzha-Stross cannot be excluded or confirmed. Doctors have to remember possibility at patients of an eosinophilia, vascular rash, deterioration in pulmonary symptoms, cardiological complications and/or neuropathy. Patients who had these symptoms it is necessary to survey and revise their scheme of treatment repeatedly.
Montelukast does not block the bronkhospastichesky answer to aspirin or to NPVS at the asthmatic patients sensitive to aspirin. Such patients have to continue to avoid intake of aspirin and various NPVS. The caution has to be observed when using montelukast together with bronchodilatory therapy. When the clinical answer is obvious, bronchodilatory therapy has to be reduced.
Use in pediatric practice
This dosage form is not appointed to children up to 15 years
of Feature of influence of medicine on ability to run the vehicle or potentially dangerous
Montiget mechanisms the car does not affect ability of the patient of a message or to operate various mechanisms. However, seldom or never, reported about drowsiness emergence therefore it is necessary to be careful when performing these types of activity.
Overdose
Symptoms: the abdominal pain, drowsiness, thirst, a headache, vomiting or psychomotor hyperactivity are possible.
Treatment: symptomatic. There are no data on a possibility of removal of montelukast by peritoneal dialysis or a hemodialysis.
A form of release and packing
On 7 tablets in blister strip packaging from aluminum foil. On the 2nd blister strip packagings of the blister together with the instruction for medical use in the state and Russian languages place in a cardboard box.
To Store storage conditions in the dry and protected from light place, at
a temperature from 15 of 0C up to 30 0C.
To store in the inaccessible place for children!
3 years
not to apply a period of storage on expiry date.
Prescription status
According to the prescription
the Producer Getz of Pharm (Pvt.) Limited, Pakistan,
29-30/27, K. I.A., Karachi 74900.
Ph. + (92-21) 111,111,511
Fax: + (92-21) 350 5792
www.getzpharma.com
Owner of the registration certificate
Getz of Pharm (Pvt.) Limited, Pakistan.
The address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products:
Representative office Getz of Pharm (Pvt.) Limited in
Almaty, Shevchenko St., 148 RK.
ph. +7 (727) 378-51-89, 378-54-78
www.getzpharma.com
To develop