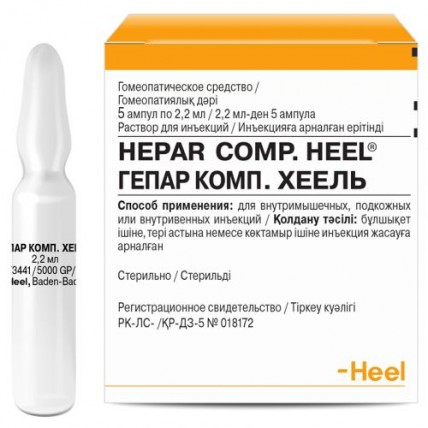
Hepar Comp. Heel 5's 2.2 ml solution for injection in ampoules
- $43.30
Sku:
8fa65b906606
Brand:
Heel (Germany)
Instruction for medical use of COMPUTER GEPAR medicine. HEEL Trade name GEPAR KOMP. HEEL the International unlicensed name Is not present the Dosage form Solution for injections of 2.2 g Structure One ampoule (2.2 g) contains active agents: Acidum alpha-ketoglutaricum D10 22.0 Acidum fumaricum D10 22.0 Acidum malicum D10 22.0 Acidum oroticum monohydricum D6 1 22.0 Acidum alpha-liponicum D8 1 22.0 Avena sativa D6 22.0 Calcium carbonicum Hahnemanni D28 22.0 Chelidonium majus D5 22.0 Cinchona pubescens D4 22.0 Colon suis D10 22.0 Cyanocobalaminum D4 22.0 Cynara scolymus D6 22.0 Duodenum suis D10 22.0 Hepar suis D8 22.0 Histaminum D10 22.0 Lycopodium clavatum D4 22.0 Natrium diethyloxalaceticum D10 22.0 Pankreas suis D10 22.0 Silybum marianum D3 22.0 Sulfur D13 22.0 Taraxacum officinale D4 22.0 Thymus suis D10 22.0 Veratrum album D4 22.0 Vesica fellea suis D10 22.0 excipients: sodium chloride, water for injections. The description Transparent solution from colourless till slightly pink color, flavourless. The pharmacotherapeutic Combination of Drugs group for treatment of diseases of a liver and biliary tract. The ATX A05C code the Pharmacological Pharmacokinetics to Investigate properties pharmacokinetic properties of homeopathic medicines is not possible. The pharmacodynamics Drug restores disintoxication function of a liver, possesses the hepatoprotective, bile-expelling, regenerating, metabolic and antioxidant action which is based on activation of protective forces of an organism and normalization of the broken functions at the expense of the substances of plant, mineral and animal origin which are a part of drug. Indications - acute and chronic diseases of a liver and biliary tract - disturbances of functions of a liver in various somatopathies and also owing to long influence of various toxic factors the Route of administration and doses the Standard dose: Adults (and children of 12 years are also more senior): On 1 ampoule from 1 to 3 times a week. Children: From 2 to 5 years: ½ ampoules (1.1 ml) from 1 to 3 times a week. From 6 to 11 years: 2/3 ampoules (1.46 ml) from 1 to 3 times a week. Route of administration: Gepar Heel compositum, solution for injections, enter intramusculary, subcutaneously or intravenously. The course of treatment makes from 3 to 6 weeks. IInstruktsiya on opening of a glass ampoule: To open an ampoule it is not necessary to make an incision it. You hold an ampoule at an angle, marking in the form of a point up, shake or knock on it until liquid appears in the lower part. Break off an ampoule head by pressing aside, opposite to marking. Side effects Seldom - reactions of hypersensitivity, such as skin allergic reactions, Contraindication fever - hypersensitivity to drug components, including to Taraxacum officinale, or to other plants from family margaritkovy Medicinal interactions of Interaction with other medicines are not established to Cynara scolymus. The special instructions Pregnancy and the period of a lactation Because of the maintenance of components of a celandine (Chelidonium) and a false hellebore (Veratrum) it is not recommended to use drug during pregnancy and a lactation. Use in pediatrics As exists insufficient experience of use of drug for children, drug should be used at children aged from 2 up to 12 years only after consultation with the doctor. The feature of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms does not affect ability to run the vehicle or potentially dangerous mechanisms. Overdose Cases of overdose of drug are not described. A form of release and packing On 2.2 ml in ampoules of colourless glass, in an upper part of an ampoule – a red dot. On 5 ampoules in blister strip packaging from PVC. Blister strip packaging together with the instruction for medical use in the state and Russian languages is put in a pack from cardboard. To Store storage conditions in original packing, at a temperature from 9 °C to 29 °C. To store out of children's reach! Not to apply a period of storage of 5 years after the expiry date specified on packing! Prescription status According to the prescription of Proizvoditel Biologishe Haylmittel Heel GmbH Dr. Rekeveg strasse 2-4, 76532 Baden-Baden, Germany the Owner of the registration certificate of Biologishe Haylmittel Heel GmbH Dr. Rekeveg strasse 2-4, 76532 Baden-Baden, Germany the Address of the organization in the territory of the Republic of Kazakhstan: the accepting claim (offer) on quality of medicines from consumers, responsible for post-registration observation of safety of medicine of AEM Services LLP 050040, RK, Almaty, Al-Farabi Ave., 75A, office 102-103 of aemservices@mail.ru +7 707 798 83 83