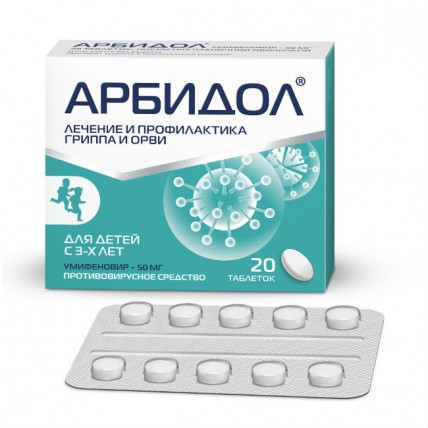
Arbidol® (Umifenovir) 50 mg, 20 tabs
- $28.60
Out Of Stock
Arbidol composition
One tablet contains the active ingredient Umifenovir (Umifenovirum) hydrochloride monohydrate - 51.75 mg (in terms of umifenovir hydrochloride - 50.00 mg);
Pharmacological properties
Pharmacodynamics. Antiviral agent. Specifically inhibits in vitro influenza A and B viruses ( Influenzavirus A , B ), including highly pathogenic subtypes A ( H 1 N 1) pdm 09 and A ( H 5 N 1), as well as other viruses that cause acute respiratory viral infections (ARVI) (coronavirus (C oronavirus ) associated with severe acute respiratory syndrome (SARS), rhinovirus ( Rhinovirus ), adenovirus ( Adenovirus ), respiratory syncytial virus ( Pneumovirus ) and parainfluenza virus ( Paramyxovirus )).
According to the mechanism of antiviral action, it belongs to fusion inhibitors (fusion), interacts with the hemagglutinin of the virus and prevents the fusion of the lipid membrane of the virus and cell membranes.
Has a moderate immunomodulatory effect, increases the body's resistance to viral infections. It has interferon-inducing activity - in a study on mice, the induction of interferons was noted already after 16 hours, and high titers of interferons remained in the blood up to 48 hours after administration. Stimulates cellular and humoral reactions of immunity: increases the number of lymphocytes in the blood, especially T cells (C D 3), increases the number of T-helpers ( CD 4), without affecting the level of T-suppressors ( CD8), normalizes the immunoregulatory index, stimulates the phagocytic function of macrophages and increases the number of natural killer cells ( NK cells).
Therapeutic efficacy for viral infections is manifested in a decrease in the duration and severity of the course of the disease and its main symptoms, as well as in a decrease in the incidence of complications associated with a viral infection and exacerbations of chronic bacterial diseases.
Refers to low-toxic drugs ( LD 50> 4 g / kg). Does not have any negative effects on the human body when taken orally in recommended doses.
Pharmacokinetics. It is quickly absorbed and distributed to organs and tissues. The maximum concentration in blood plasma when taken at a dose of 50 mg is achieved after 1.2 hours, at a dose of 100 mg - after 1.5 hours. It is metabolized in the liver. The half-life is 17-21 hours. About 40% is excreted unchanged, mainly with bile (38.9%) and in a small amount by the kidneys (0.12%). During the first day, 90% of the administered dose is removed.
Indications for use:
- Prevention and treatment in adults and children: influenza A and B, other acute respiratory viral infections.
- Complex therapy of acute intestinal infections of rotavirus etiology in children over 3 years old.
- Complex therapy of chronic bronchitis, pneumonia and recurrent herpes infection.
- Prevention of postoperative infectious complications.
Contraindications
Hypersensitivity to umifenovir or any component of the drug, children under 3 years of age. First trimester of pregnancy.
With care
Second and third trimesters of pregnancy.
Application during pregnancy and during breastfeeding
In animal studies, no harmful effects on the course of pregnancy, the development of the embryo and fetus, labor and postnatal development were found.
The use of the drug Arbidol® in the first trimester of pregnancy is contraindicated. In the second and third trimester of pregnancy, Arbidol® can only be used for the treatment and prevention of influenza and if the intended benefit to the mother outweighs the potential risk to the fetus. The benefit / risk ratio is determined by the attending physician.
It is not known whether Arbidol® passes into breast milk in women during lactation. If it is necessary to use Arbidol®, breastfeeding should be stopped.
Method of administration and dosage
Inside, before meals.
Single dose (depending on age):
Age | Single dose of the drug |
| from 3 to 6 years old | 50 mg (1 tablet) |
| from 6 to 12 years old | 100 mg (2 tablets) |
| over 12 years old and adults | 200 mg (4 tablets) |
Indication | The scheme of taking the drug |
| In children from 3 years of age and adults: | |
| Non-specific prophylaxis during an epidemic of influenza and other acute respiratory viral infections | in a single dose 2 times a week for 3 weeks. |
| Non-specific prophylaxis through direct contact with patients with influenza and other acute respiratory viral infections | in a single dose 1 time per day for 10-14 days. |
| Treatment of influenza and other acute respiratory viral infections | in a single dose 4 times a day (every 6 hours) for 5 days. |
| In children from 3 years old: | |
| Complex therapy of acute intestinal infections of rotavirus etiology | in a single dose 4 times a day (every 6 hours) for 5 days. |
| In children from 3 years of age and adults: | |
| Complex therapy of chronic bronchitis, pneumonia, herpes infection | in a single dose 4 times a day (every 6 hours) for 5-7 days, then a single dose 2 times a week for 4 weeks. |
| Prevention of postoperative infectious complications | in a single dose 2 days before surgery, then 2 and 5 days after surgery. |
Taking the drug begins from the moment the first symptoms of influenza and other acute respiratory viral infections appear, preferably no later than 3 days from the onset of the disease.
If after taking the drug Arbidol® for three days in the treatment of influenza and other acute respiratory viral infections, the severity of the symptoms of the disease, including high temperature (38 ° C or more), persists, then you should consult a doctor to assess the validity of taking the drug.
Use the drug only according to the indications, the method of administration and the doses indicated in the instructions.
In the treatment of influenza and acute respiratory viral infections, concomitant symptomatic therapy is possible, including taking antipyretic drugs, mucolytic and local vasoconstrictors.
Side effect
The drug Arbidol® belongs to low-toxic drugs and is usually well tolerated.
Side effects are rare, usually mild to moderate, and transient.
The incidence of adverse drug reactions is determined in accordance with the WHO classification: very often (with a frequency of more than 1/10), often (with a frequency of at least 1/100, but less than 1/10), infrequently (with a frequency of at least 1/1000, but less than 1/100), rarely (with a frequency of not less than 1/10000, but less than 1/1000), very rarely (with a frequency of less than 1/10000), the frequency is unknown (cannot be established from the available data)
Immune system disorders: rarely - allergic reactions.
If any of the side effects indicated in the instructions are aggravated, or you notice any other side effects not listed in the instructions, inform your doctor.
Overdose
Not checked.
Interaction with other medicinal products
When administered with other drugs, no negative effects were noted.
Special clinical studies devoted to the study of the interaction of the drug Arbidol® with other drugs have not been conducted.
Information about the presence of undesirable interactions with antipyretic, mucolytic and local vasoconstrictor drugs in the conditions of a clinical study was not identified.
Special instructions
It is necessary to follow the recommended scheme and duration of the drug intake. In case of missing one dose of the drug, the missed dose should be taken as early as possible and the course of taking the drug should be continued according to the started scheme. If after taking the drug Arbidol® for three days in the treatment of influenza and other acute respiratory viral infections, the severity of the symptoms of the disease, including high temperature (38 ° C or more), persists, then you should consult a doctor to assess the validity of taking the drug.
Influence on the ability to drive vehicles and mechanisms
Does not exhibit central neurotropic activity and can be used in medical practice in people of various professions, incl. requiring increased attention and coordination of movements (transport drivers, operators, etc.).
Shelf life 3 years.
Do not use after the expiration date printed on the package.
Storage conditions
At a temperature not exceeding 25 ° C. Keep out of the reach of children.