-30%
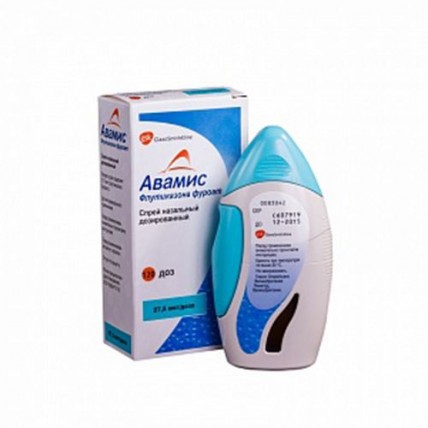
Avamys® 120 doses Nasal Spray
- $32.00
- $46.00
Avamys nasal spray is used to treat symptoms of allergic rhinitis including stuffy, runny or itchy nose, sneezing and watery, itchy or red eyes, in adults and children aged 6 years and over. Allergy symptoms can occur at specific times of the year and be caused by allergy to pollen from grass or trees (hayfever), or they can occur all year round and be caused by allergy to animals, house-dust mites or moulds. Avamys belongs to a group of medicines called glucocorticoids. Avamys works to decrease inflammation caused by allergy (rhinitis).
Common brand names: Flonase (Fluticasone Propionate), XHANCE® (fluticasone propionate), Cutivate® (fluticasone topical), Veramyst (Fluticasone Furoate), ARNUITY® ELLIPTA® (fluticasone furoate), BREO ELLIPTA (Fluticasone Furoate/Vilanterol), Relvar Ellipta (Fluticasone Furoate/Vilanterol)
1 dose contains the active ingredient - fluticasone furoate (micronized) 27.5 mcg
Treatment of symptoms of allergic rhinitis in adults and children over 6 years of age
Avamis is intended for intranasal administration only.
To obtain complete therapy, the drug is recommended to be used according to the schedule described below. The onset of action of the drug is observed within 8 hours after the first injection, however, it may take several days before reaching the maximum effect, which should be warned the patient. The duration of therapy should be limited to the period of allergic exposure.
Adults and adolescents over 12 years old
The initial recommended dose is two injections into each nasal passage once a day (total daily dose 110 mcg).
As the symptoms of the disease are controlled, the dose is reduced to 1 injection into each nasal passage once a day (55 mcg per day).
The dose should be titrated to the lowest dose that achieves symptom control.
Children from 6 to 11 years old
One injection (27.5 mcg) into each nasal passage once a day (55 mcg per day). In the absence of an effect with this dosing regimen, the daily dose of the drug can be increased to 110 mcg per day, but the daily dose should not exceed 2 injections into each nasal passage. As the symptoms of the disease are controlled, the dose is reduced to 1 injection into each nasal passage once a day (55 mcg per day).
Children under 6 years old
The experience of this category of persons is limited. There are no data on efficacy and safety in this cohort of individuals.
The elderly
No dose adjustment is required.
Application for renal failure
No dose adjustment is required.
Application for liver failure
No dose adjustment is required for mild to moderate hepatic impairment. There is no data on the use of the drug in severe hepatic insufficiency, but caution should be exercised when using it, since this cohort of individuals has a greater risk of developing systemic adverse reactions associated with the use of corticosteroids.
Avamis is in a glass bottle in a plastic case.
The case has a window allowing you to see the remainder of the drug in the vial. When the button is firmly pressed, the suspension is sprayed through the tip. The tip is protected by a cap.
Checking when using Avamis
Before use, you should gently shake the bottle, take it by placing your index and middle fingers on either side of the tip, and your thumb under the bottom.
When using the drug for the first time, check that the nebulizer is working properly Aim the tip away from you, press at least 6 times until a small cloud appears from the tip. The device should be re-checked only if the vial has been stored without a cap for 5 days and if the drug has not been used for 30 days.
How to use Avamys spray
You need to clear your nose (blow your nose slightly). Close one nasal passage and insert the tip into the other nasal passage. Tilt your head slightly forward while continuing to hold the bottle upright. Then you should begin to inhale through the nose and, while continuing to inhale, press the button once with your finger to spray the drug. Exhale through your mouth. Repeat the procedure for a second spray into the same nasal passage, if necessary. Then completely repeat the described procedure, introducing the tip into the other nasal passage.
After use, blot the tip and button with a dry, clean cloth. Put on the protective cap. Do not wash in water.
The adverse reactions listed below are grouped by frequency of occurrence: very frequent (> 1/10), frequent (> 1/100, <1/10), infrequent (> 1/1000, <1/100), rare (≥1 / 10,000 and <1/1000), very rare (<1/10,000).
Very Often
- nosebleeds
In adults and adolescents, nosebleeds are more common with prolonged use of the drug (more than 6 weeks) than with short-term use. In children, there was no association between bleeding and the duration of therapy.
Often
- headache
- ulceration of the nasal mucosa
Infrequently
- pain, increased sensitivity, dryness, burning, irritation in the nasal passages, discomfort, including nosebleeds
Rarely
- hypersensitivity reactions, including anaphylactic reactions, vascular edema, rash and urticaria
Unknown
- growth retardation in children (in a one-year clinical study in prepubertal children receiving Avamis at a dose of 110 mcg per day, the average growth retardation was -0.27 cm compared with the placebo group)
- hypersensitivity to any of the components of the drug
- combined use with ritonavir
- children under 6 years old
Care should be taken when using Avamis simultaneously with strong inhibitors of the CYP3A4 enzyme. Concomitant use with such an inhibitor of the CYP3A4 isoenzyme as ritonavir is not recommended due to the potential risk of increased systemic excretion of fluticasone furoate.
The CYP3A4 inhibitor ketoconazole may increase the plasma concentration of fluticasone (in clinical studies, when compared in the ketoconazole group, there was a more frequent increase in plasma fluticasone levels (6 out of 20 patients) than in the placebo group (1 out of 20), which did not generally affect the difference in the concentration of fluticasone with 24 hour monitoring of the two groups).
There is no reason to assume any metabolic interactions and inhibitory actions between Avamis and the metabolism of other drugs mediated by cytochrome P450, but no clinical studies have been conducted to study this interaction.
The drug is indicated for intranasal use only. For full therapeutic effect, a regular treatment regimen is recommended. It may take several days to achieve maximum effect.
A systemic effect of the drug is possible, mainly when using doses exceeding the recommended ones, as well as with prolonged use of the drug. Systemic effects are much lower than with oral corticosteroids and may differ from patient to patient. Potential systemic effects may include Cushing's syndrome, development of Cushingoid symptoms, suppression of adrenal function, stunted growth in children and adolescents, cataracts, glaucoma, and, in more rare cases, psychological and behavioral effects (psychomotor hyperreactivity, sleep disturbance, anxiety, depression, aggression - mostly in children).
The use of the drug in doses higher than recommended, can lead to clinically significant suppression of adrenal function. In case of development of symptoms characteristic of adrenal suppression, the use of systemic corticosteroids should be considered during periods of stress or surgery. The use of Avamis at a dose of 110 mcg per day did not cause disturbances in the hypothalamic-pituitary-adrenal system in adults, children and adolescents, nevertheless, once control over the symptoms of rhinitis is achieved, the dose should be reduced to the minimum effective dose. As with other intranasal corticosteroids, it is necessary to assess the total systemic load with the concomitant use of other forms of corticosteroids.
If there are suggestions of impaired adrenal function, care must be taken when transferring patients from treatment with systemic corticosteroids to Avamis.
Intranasal and inhaled corticosteroids can cause glaucoma and / or cataracts. Careful monitoring of patients with complaints of changes in visual function or with increased intraocular pressure, glaucoma and / or cataract in history is required.
Cases of growth retardation in children who received the drug at the recommended doses (110 mcg per day) were identified, and therefore it is recommended to regularly monitor the growth of children on long-term therapy with intranasal corticosteroids. If the fact of growth retardation is revealed, the ongoing therapy should be revised in order to reduce the dose received to the minimum effective dose. It is also necessary to consult a pediatrician.
Avamis contains benzalkonium chloride, which can irritate the nasal mucosa. Co-administration with ritonavir is not recommended due to the increased risk of systemic exposure to fluticasone furoate.
Renal dysfunction
Fluticasone furoate is not detected in the urine of healthy people after intranasal administration. Less than 1% is excreted in the urine, and therefore renal impairment does not affect the pharmacokinetics of fluticasone furoate.
Liver dysfunction
An average dose of 110 mcg in patients with moderate hepatic impairment does not suppress cortisol production. In patients with severe hepatic impairment, caution should be exercised when prescribing Avamis due to the possibility of more frequent development of systemic adverse reactions.
Pregnancy and lactation
When using the drug at a dose of 110 μg per day, the plasma concentration of fluticasone furoate was undetectable, and therefore the potential risk of toxic effects on the fetus is very low.
However, the use of Avamis during pregnancy is possible only if the expected benefit to the mother outweighs the potential risk to the fetus.
It is not known whether fluticasone is excreted in breast milk, therefore, the use of Avamis is possible only when the expected benefit to the mother outweighs the potential risk to the child.
Features of the effect of the drug on the ability to drive a vehicle or potentially dangerous mechanisms
Special studies have not been conducted, the effect of the drug on the ability to administer expected to be a vehicle or potentially dangerous machinery.
Symptoms: when using doses exceeding the recommended dose for adults 24 times within 3 days, no systemic adverse reactions were observed.
Treatment: in case of acute overdose, the need for any therapy other than observation is unlikely.
Store at a temperature not exceeding 30 ° C. Do not freeze.
Store no more than 2 months after the first opening.
Keep out of the reach of children!
Shelf life - 3 years
Do not take the drug after the expiration date.
Common brand names: Flonase (Fluticasone Propionate), XHANCE® (fluticasone propionate), Cutivate® (fluticasone topical), Veramyst (Fluticasone Furoate), ARNUITY® ELLIPTA® (fluticasone furoate), BREO ELLIPTA (Fluticasone Furoate/Vilanterol), Relvar Ellipta (Fluticasone Furoate/Vilanterol)
Composition
1 dose contains the active ingredient - fluticasone furoate (micronized) 27.5 mcg
Indications for use
Treatment of symptoms of allergic rhinitis in adults and children over 6 years of age
Method of administration and dosage
Avamis is intended for intranasal administration only.
To obtain complete therapy, the drug is recommended to be used according to the schedule described below. The onset of action of the drug is observed within 8 hours after the first injection, however, it may take several days before reaching the maximum effect, which should be warned the patient. The duration of therapy should be limited to the period of allergic exposure.
Adults and adolescents over 12 years old
The initial recommended dose is two injections into each nasal passage once a day (total daily dose 110 mcg).
As the symptoms of the disease are controlled, the dose is reduced to 1 injection into each nasal passage once a day (55 mcg per day).
The dose should be titrated to the lowest dose that achieves symptom control.
Children from 6 to 11 years old
One injection (27.5 mcg) into each nasal passage once a day (55 mcg per day). In the absence of an effect with this dosing regimen, the daily dose of the drug can be increased to 110 mcg per day, but the daily dose should not exceed 2 injections into each nasal passage. As the symptoms of the disease are controlled, the dose is reduced to 1 injection into each nasal passage once a day (55 mcg per day).
Children under 6 years old
The experience of this category of persons is limited. There are no data on efficacy and safety in this cohort of individuals.
The elderly
No dose adjustment is required.
Application for renal failure
No dose adjustment is required.
Application for liver failure
No dose adjustment is required for mild to moderate hepatic impairment. There is no data on the use of the drug in severe hepatic insufficiency, but caution should be exercised when using it, since this cohort of individuals has a greater risk of developing systemic adverse reactions associated with the use of corticosteroids.
Instructions for use
Avamis is in a glass bottle in a plastic case.
The case has a window allowing you to see the remainder of the drug in the vial. When the button is firmly pressed, the suspension is sprayed through the tip. The tip is protected by a cap.
Checking when using Avamis
Before use, you should gently shake the bottle, take it by placing your index and middle fingers on either side of the tip, and your thumb under the bottom.
When using the drug for the first time, check that the nebulizer is working properly Aim the tip away from you, press at least 6 times until a small cloud appears from the tip. The device should be re-checked only if the vial has been stored without a cap for 5 days and if the drug has not been used for 30 days.
How to use Avamys spray
You need to clear your nose (blow your nose slightly). Close one nasal passage and insert the tip into the other nasal passage. Tilt your head slightly forward while continuing to hold the bottle upright. Then you should begin to inhale through the nose and, while continuing to inhale, press the button once with your finger to spray the drug. Exhale through your mouth. Repeat the procedure for a second spray into the same nasal passage, if necessary. Then completely repeat the described procedure, introducing the tip into the other nasal passage.
After use, blot the tip and button with a dry, clean cloth. Put on the protective cap. Do not wash in water.
Side effects
The adverse reactions listed below are grouped by frequency of occurrence: very frequent (> 1/10), frequent (> 1/100, <1/10), infrequent (> 1/1000, <1/100), rare (≥1 / 10,000 and <1/1000), very rare (<1/10,000).
Very Often
- nosebleeds
In adults and adolescents, nosebleeds are more common with prolonged use of the drug (more than 6 weeks) than with short-term use. In children, there was no association between bleeding and the duration of therapy.
Often
- headache
- ulceration of the nasal mucosa
Infrequently
- pain, increased sensitivity, dryness, burning, irritation in the nasal passages, discomfort, including nosebleeds
Rarely
- hypersensitivity reactions, including anaphylactic reactions, vascular edema, rash and urticaria
Unknown
- growth retardation in children (in a one-year clinical study in prepubertal children receiving Avamis at a dose of 110 mcg per day, the average growth retardation was -0.27 cm compared with the placebo group)
Contraindications
- hypersensitivity to any of the components of the drug
- combined use with ritonavir
- children under 6 years old
Drug interactions
Care should be taken when using Avamis simultaneously with strong inhibitors of the CYP3A4 enzyme. Concomitant use with such an inhibitor of the CYP3A4 isoenzyme as ritonavir is not recommended due to the potential risk of increased systemic excretion of fluticasone furoate.
The CYP3A4 inhibitor ketoconazole may increase the plasma concentration of fluticasone (in clinical studies, when compared in the ketoconazole group, there was a more frequent increase in plasma fluticasone levels (6 out of 20 patients) than in the placebo group (1 out of 20), which did not generally affect the difference in the concentration of fluticasone with 24 hour monitoring of the two groups).
There is no reason to assume any metabolic interactions and inhibitory actions between Avamis and the metabolism of other drugs mediated by cytochrome P450, but no clinical studies have been conducted to study this interaction.
Special instructions
The drug is indicated for intranasal use only. For full therapeutic effect, a regular treatment regimen is recommended. It may take several days to achieve maximum effect.
A systemic effect of the drug is possible, mainly when using doses exceeding the recommended ones, as well as with prolonged use of the drug. Systemic effects are much lower than with oral corticosteroids and may differ from patient to patient. Potential systemic effects may include Cushing's syndrome, development of Cushingoid symptoms, suppression of adrenal function, stunted growth in children and adolescents, cataracts, glaucoma, and, in more rare cases, psychological and behavioral effects (psychomotor hyperreactivity, sleep disturbance, anxiety, depression, aggression - mostly in children).
The use of the drug in doses higher than recommended, can lead to clinically significant suppression of adrenal function. In case of development of symptoms characteristic of adrenal suppression, the use of systemic corticosteroids should be considered during periods of stress or surgery. The use of Avamis at a dose of 110 mcg per day did not cause disturbances in the hypothalamic-pituitary-adrenal system in adults, children and adolescents, nevertheless, once control over the symptoms of rhinitis is achieved, the dose should be reduced to the minimum effective dose. As with other intranasal corticosteroids, it is necessary to assess the total systemic load with the concomitant use of other forms of corticosteroids.
If there are suggestions of impaired adrenal function, care must be taken when transferring patients from treatment with systemic corticosteroids to Avamis.
Intranasal and inhaled corticosteroids can cause glaucoma and / or cataracts. Careful monitoring of patients with complaints of changes in visual function or with increased intraocular pressure, glaucoma and / or cataract in history is required.
Cases of growth retardation in children who received the drug at the recommended doses (110 mcg per day) were identified, and therefore it is recommended to regularly monitor the growth of children on long-term therapy with intranasal corticosteroids. If the fact of growth retardation is revealed, the ongoing therapy should be revised in order to reduce the dose received to the minimum effective dose. It is also necessary to consult a pediatrician.
Avamis contains benzalkonium chloride, which can irritate the nasal mucosa. Co-administration with ritonavir is not recommended due to the increased risk of systemic exposure to fluticasone furoate.
Renal dysfunction
Fluticasone furoate is not detected in the urine of healthy people after intranasal administration. Less than 1% is excreted in the urine, and therefore renal impairment does not affect the pharmacokinetics of fluticasone furoate.
Liver dysfunction
An average dose of 110 mcg in patients with moderate hepatic impairment does not suppress cortisol production. In patients with severe hepatic impairment, caution should be exercised when prescribing Avamis due to the possibility of more frequent development of systemic adverse reactions.
Pregnancy and lactation
When using the drug at a dose of 110 μg per day, the plasma concentration of fluticasone furoate was undetectable, and therefore the potential risk of toxic effects on the fetus is very low.
However, the use of Avamis during pregnancy is possible only if the expected benefit to the mother outweighs the potential risk to the fetus.
It is not known whether fluticasone is excreted in breast milk, therefore, the use of Avamis is possible only when the expected benefit to the mother outweighs the potential risk to the child.
Features of the effect of the drug on the ability to drive a vehicle or potentially dangerous mechanisms
Special studies have not been conducted, the effect of the drug on the ability to administer expected to be a vehicle or potentially dangerous machinery.
Overdose
Symptoms: when using doses exceeding the recommended dose for adults 24 times within 3 days, no systemic adverse reactions were observed.
Treatment: in case of acute overdose, the need for any therapy other than observation is unlikely.
Storage conditions
Store at a temperature not exceeding 30 ° C. Do not freeze.
Store no more than 2 months after the first opening.
Keep out of the reach of children!
Shelf life - 3 years
Do not take the drug after the expiration date.