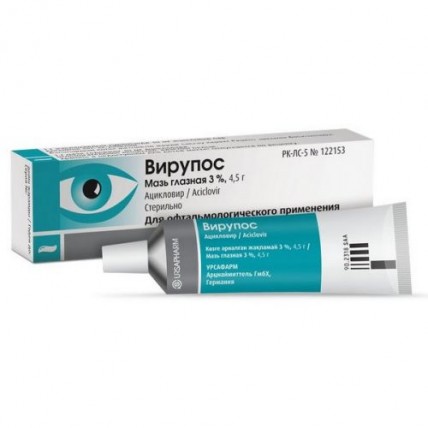
VIRUPOS (Acyclovir) Ophthalmic Ointment 3% , 4.5 g
- $21.00
What is VIRUPOS Ophthalmic Ointment?
VIRUPOS Ophthalmic Ointment is an antiviral eye medication containing 3% Acyclovir, specifically designed to treat eye infections caused by the herpes simplex virus. It is a smooth ointment ranging in color from white to grayish-white, and it is applied directly to the eye to inhibit the replication of viral DNA. This treatment is highly effective for both superficial and internal eye infections and provides targeted relief without significant systemic absorption.
Stored in a convenient 4.5 g aluminum tube, VIRUPOS is a trusted solution for managing herpes-related eye conditions.
How Does VIRUPOS Work?
VIRUPOS contains the active ingredient Acyclovir, which penetrates infected cells to combat the herpes simplex and varicella-zoster viruses. Once inside the infected cell, Acyclovir is converted into its active form, disrupting the virus's DNA synthesis. This stops the virus from replicating and helps the immune system eliminate the infection.
When applied to the eye, the ointment achieves high concentrations of the active ingredient in the aqueous humor, effectively treating both surface and internal infections caused by the herpes virus.
What Are the Indications for Using VIRUPOS?
VIRUPOS is primarily used for the treatment of keratitis, a condition characterized by inflammation of the cornea, caused by the herpes simplex virus.
How Should You Apply VIRUPOS?
- Wash your hands thoroughly before application.
- Squeeze a strip of ointment approximately 1 cm long into the lower conjunctival sac of the affected eye.
- Apply the ointment 5 times a day at four-hour intervals.
- After applying, gently close your eyelids and rotate your eyeballs to spread the ointment evenly.
Continue treatment for at least 3 days after the corneal lesion has healed. Avoid touching the tube's tip to the eye or any other surface to maintain sterility.
What Are the Possible Side Effects of VIRUPOS?
Side effects are typically mild and temporary. They include:
- Very common: Superficial punctate keratopathy (does not require discontinuation and resolves without complications).
- Common: Temporary mild burning or stinging sensation in the eye, conjunctivitis.
- Rare: Blepharitis (inflammation of the eyelids).
- Very rare: Hypersensitivity reactions, including angioedema and urticaria (hives).
If you experience severe or persistent symptoms, consult your healthcare provider.
Are There Any Precautions When Using VIRUPOS?
- Do not wear contact lenses during treatment with VIRUPOS.
- Inform patients about potential temporary blurred vision immediately after application, which may impair driving or operating machinery.
- Pregnant or breastfeeding women should only use VIRUPOS if the potential benefits outweigh the risks. While systemic absorption is minimal, caution is advised.
How Should You Store VIRUPOS?
- Store the ointment at a temperature not exceeding 25°C (77°F).
- Keep the product out of reach of children.
- After opening, use the product within 1 month.
Do not use the ointment past the expiration date printed on the packaging.
Why Choose VIRUPOS?
VIRUPOS Ophthalmic Ointment is a proven, effective, and safe antiviral treatment for herpes-related eye infections.
Its targeted action ensures high efficacy with minimal systemic absorption, making it suitable for most patients when used as directed.
By following the correct application methods and precautions, users can achieve optimal results and relief from discomfort caused by keratitis.