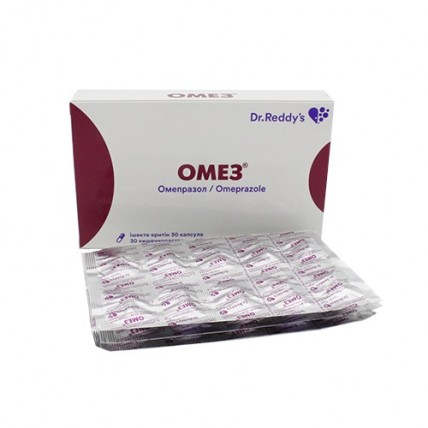
Omez (Omeprazole) 20 mg, 30 capsules
- $12.80
The instruction for medical use
of OMEZ® medicine
the Trade name
of Omez®
the International unlicensed
name Omeprazolum Dosage Form
of the Capsule kishechnorastvorimy 20 mg
One capsule contains Structure:
active agent - omeprazolum of 20 mg,
excipients: Mannitolum, lactose, sodium lauryl sulfate, dinatrium hydrophosphate anhydrous, sucrose, sucrose (25/30), gipromelloza (6cps)
main cover: gipromelloza (6 cps)
kishechnorastvorimy cover: methacrylic acid a kopolimer (type C) (eudragit L 100-55), sodium hydroxide, a macrogoal 6000, talc purified the titan E171 dioxide
structure of the capsule:
body: gelatin, methylparaben, propylparaben, sodium lauryl sulfate, E122 carmoisin,
lid: gelatin, methylparaben, propylparaben, sodium lauryl sulfate, karmoizin E122
the Description
Solid gelatin capsules No. 2 with the colourless transparent body and a transparent pink lid, with the inscription OMEZ on the body and a lid with the double blocking system. Contents of capsules – spherical pellets of white or almost white color.
Pharmacotherapeutic group
Antiulcerous drugs and drugs for treatment of a gastroegzofagealny reflux. Inhibitors of the protonew pump. Omeprazolum.
The ATX A02BC01 code
the Pharmacological
Pharmacokinetics Omeprazolum properties is put into acid resisting pellets which are dissolved only in intestines. At intake it is quickly absorbed from digestive tract. Meal does not affect bioavailability of drug. At reception of 20-40 mg of omeprazolum the bioavailability is about 40%, after repeated reception increases bioavailability about 60%. The peak of concentration of omeprazolum in blood plasma is reached within 0.5 - 3.5 hours. Communication with proteins of plasma - about 97%. Omeprazolum is metabolized in a liver. Metabolism is carried out through the system of P450 cytochrome (CYP2C19 isoform - S-Mephenytoinum a hydroxylase). Concentration of omeprazolum in plasma does not correlate with degree and duration of suppression of secretion of Acidum hydrochloricum (t½ - 0.5-1 hour, and the anti-secretory effect lasts up to 2-3 days).
The most part of drug of nearly 80% is removed by kidneys in the form of metabolites. Elimination half-life makes 0.5-1 hour.
The pharmacodynamics
Omeprazolum belongs to benzimidazolny derivatives. In acidic environment of covering cells of a stomach omeprazolum turns into an active metabolite - sulphenamide which it is irreversible contacts one of the N+,k cysteic groups +-ATF-azy and inhibits activity of enzyme (proton pomp). Thus, omeprazolum interferes with process of a kislotoobrazovaniye at the level of a final stage of synthesis of hydrochloric acid that leads to decrease in level of basal and stimulated secretion, irrespective of the irritant nature.
The inhibition of activity of enzyme has dose-dependent character.
Indications
Adults
- treatment of an ulcer of a duodenum, prevention of a recurrence of ulcers of a duodenum
- treatment of stomach ulcer, prevention of a recurrence of stomach ulcer
- in combination with the corresponding antibiotics, Helicobacter Pylori (H.P) eradication in a peptic ulcer
- treatment connected with intake of non-steroidal anti-inflammatory drugs (NPVP) - associated from gastropathies of a stomach and duodenum, prevention the gastropathies of a stomach and a duodenum connected with reception NPVP-assotsiirovannoy at patients with risk
- treatment a reflux esophagitis, long-term treatment of patients about a reflux esophagitis
- symptomatic treatment of a gastroesophagal reflux
- treatment of a syndrome
of Zollingera-Ellison Primeneniye in pediatrics
Children are more senior than 1 year and ≥ 10 kg
- treatment a reflux esophagitis
- symptomatic treatment of heartburn and an eructation acid in gastroesophagal a reflux
the Children's age is more senior than 4 years and teenage age
- in a combination with antibiotics in treatment of a peptic ulcer of a stomach and the duodenum caused by H. Pylori
the Route of administration and doses
Adult
the Recommended Omez's dose for treatment of a peptic ulcer of a duodenum – 20 mg (on 1 capsule) once a day within 4 weeks. Treatment can be prolonged for 4 weeks. For patients with a slabochuvstvitelny ulcer of a duodenum the use of Омез® 40 mg once a day, a course of treatment within 4 weeks is recommended.
The recommended Omez's dose for treatment of a peptic ulcer of a stomach – on 20 mg, two once a day (40 mg/days) within 4-8 weeks.
The recommended Omez's dose for a short course of treatment of a gastroesophageal reflux disease (GERD), not erosive esophagitis – 20 mg (on 1 capsule) daily once a day within 4 weeks.
The recommended Omez's dose for prevention of a recurrence of stomach ulcer - 20 mg once a day within 4 weeks. If necessary the dose of the drug Omez® can be increased up to 40 mg once a day, within 4 weeks.
For patients with an erosive esophagitis and with the complicated Omez GERD forms it is appointed in a dose of 20 mg (on 1 capsule) within 4 – 8 weeks.
In case of insufficient effectiveness of the above-stated therapy at patients with the I-II stages of GERD about 20 mg two times a day, within 4 - 8 weeks are recommended to accept, at III-IV stages of GERD recommend to accept on 40 mg once a day, within 4 - 8 weeks.
At more long-term treatment the dose of 20 mg at reception is appointed once a day, in the morning.
The recommended initial dose of Omez for the patients suffering from Zollingera-Ellison's syndrome – 60 mg. If necessary the dose is increased to 120 mg a day, dividing into 2 receptions.
Helicobacter pylori – the associated stomach diseases as a part of complex therapy: the recommended dose of 20 mg 2 times a day (it is strict in 12 hours, in the morning and in the evening) in a combination with antimicrobic therapy within 7 - 14 days.
- Omez of 20 mg + klaritromitsin 500 mg + amoxicillin of 1000 mg, two times a day within one week, or
- Omez of 20 mg + klaritromitsin 250 mg (or 500 mg) + metronidazole of 400 mg (either 500 mg or Tinidazolum of 500 mg), each drug two times a day within one week, or
- Omez of 40 mg once a day with amoxicillin of 500 mg and metronidazole of 400 mg (either 500 mg or Tinidazolum of 500 mg), both three times a day within one week.
The digestive tract erosive cankers connected with intake of non-steroidal anti-inflammatory drugs - the recommended dose - 20 mg two times (40 mg) a day within 4-8 weeks. For the purpose of prevention of appearance of new ulcers at use of non-steroidal anti-inflammatory drugs the long-term use of drug on 20 mg once a day, is recommended in the morning.
The digestive tract erosive cankers connected with intake of non-steroidal anti-inflammatory drugs - the recommended dose - 20 mg two times a day, within 4 - 8 weeks.
The recommended Omez's dose for prevention connected with intake of non-steroidal anti-inflammatory drugs - the associated stomach ulcer and a duodenum at patients of risk group (возраст>, 60, ulcer of stomach and duodenum in the anamnesis, earlier had gastrointestinal bleeding), the recommended dose - 20 mg once a day.
The recommended Omez's dose for treatment of a reflux esophagitis - 20 mg makes once a day, within 4 weeks. For patients with a heavy esophagitis the recommended Omez's dose of 40 mg once a day, within 8 weeks.
The recommended Omez's dose for treatment of a symptomatic gastroesophageal reflux - 20 mg once a day, within 4 weeks.
The recommended initial dose of Omez for the patients suffering from Zollingera-Ellison's syndrome - 60 mg once a day. If necessary the dose is increased to 120 mg a day, dividing into 2 receptions if the dose of drug exceeds 80 mg a day, it has to be divided and accepted two times a day.
Drug is recommended to be used before food, without damaging the capsule, to wash down with a small amount of liquid.
The children's age
Children is more senior than 1 year and & gt, 10 kg
Treatment of a reflux esophagitis
Symptomatic treatment of heartburn and regurgitation of acid in a gastroesophageal reflux disease, the recommended dose:
Age
Weight
Dosage
& gt, 1 year
of 10-20
kg 10 mg once a day. If necessary the dose can be increased up to 20 mg once a day
& gt, 2 years
& gt, 20
kg 20 mg once a day. If necessary the dose can be increased up to 40 mg
the Recommended Omez's dose for treatment a reflux esophagitis once a day - 20 mg, within 4 - 8 weeks.
The recommended Omez's dose at symptomatic treatment of heartburn and regurgitation of acid in a gastroesophageal reflux disease – 20 mg, within 2-4 weeks.
The children's age is more senior than 4 years and teenage age
the Recommended Omez's dose for treatment of a peptic ulcer of a stomach and a duodenum of pylori caused by N. – in courses of 1 - 2 week in a combination with antibacterial drugs:
Weight
the Dosage
of 15-30 kg
the Combination with two antibiotics: Omez of 10 mg, amoxicillin of 25 mg/kg of body weight and klaritromitsin 7.5 mg/kg of body weight are appointed in common two times a day within one week.
31-40 kg
the Combination with two antibiotics: Omez of 20 mg, amoxicillin of 750 mg and klaritromitsin 7.5 mg/kg of body weight are appointed in common two times a day within one week.
& gt, 40 kg
the Combination with two antibiotics: Omez of 20 mg, amoxicillin of 1 g and klaritromitsin 500 mg are appointed in common two times a day within one week.
The special group of the population
the Renal failure
with a renal failure dose adjustment is not required the Liver failure
At patients with a liver failure the patients can have a sufficient daily dose of 10-20 mg
Patients of advanced age (& gt, 65 years)
For patients of advanced age the dose adjustment is not required
the Route of administration
It is recommended to take the pill Omez in the morning, to swallow them entirely, washing down with a half of a glass of water.
For the patients having difficulties with swallowing and for children who can drink or swallow semi-fluid food
the Patients can dissolve it in a spoon of still water and if wish, to mix it with some fruit juice or apple puree. Patients have to be notified that dispersion has to be accepted immediately (or within 30 minutes), it should be mixed only just before the use and to wash down with a half of a glass of water. NOT to USE milk or sparkling water. Kishechnorastvorimy granules should not be chewed.
Side effects
- thorax pains, tachycardia or bradycardia, heartbeat, hypertensia, peripheral hypostases
- lack of appetite, a food faddism, an atrophy mucous language, dry language, esophageal candidiasis, a meteorism, irritation of intestines
- moderate tranzitorny increase in activity of transaminases, pancreatitis
- a hyponatremia, a hypoglycemia, increase in body weight
- spasms, muscle weakness, joint pains, the lower extremity pains
- a depression, aggression, hallucinations, drowsiness or insomnia, confusion of consciousness, nervousness, a tremor, apathy, feeling of alarm, dizziness, paresthesias
- disorders of vision, sonitus, nasal bleeding, a sore throat
- hemorrhagic rash and/or petechias, inflammation, an itching, dryness of integuments, a hyperhidrosis, an alopecia
- urticaria, an itching, a Quincke's disease
- interstitial nephrite, infections of urinary tract, a proteinuria, a hamaturia, a glucosuria
- pain in testicles, a gynecomastia
Seldom
- a neutropenia, a leukopenia, an agranulocytosis, a leukocytosis, thrombocytopenia, anemia, including hemolytic anemia, a pancytopenia
- the hepatocellular, cholestatic or mixed hepatitis
Very seldom
- a Lyell's disease, Stephens-Johnson's syndrome, a multiformny erythema
- risk of developing spinal fractures, necks of a hip,
the Contraindication wrist
- hypersensitivity to any of drug components
- simultaneous use with nelfinaviry
Medicinal interactions
of the Possibility of interaction of omeprazolum with other medicines are limited. Omeprazolum is metabolized in a liver through a system P450 cytochrome therefore at simultaneous use with drugs which are metabolized in a liver omeprazolum can reduce their removal. Omeprazolum slows down excretion of diazepam, Phenytoinum and anticoagulants (warfarin). At combined use with anticoagulants the control of blood test, a prothrombin time is necessary, correction of a dose of omeprazolum is necessary.
Nelfinavir, atazanavir
Plasma levels of a nelfinavir and atazanavir decrease in case of their combined use with omeprazolum.
Joint prescribing of omeprazolum with nelfinaviry contraindicated (see the section Simultaneous use of omeprazolum (40 mg once a day) reduces average exposure of a nelfinavir by 40%, and average exposure pharmacological of an active metabolite of M8 was reduced by 75-90%. Interaction can include CYP2C19 inhibition also.
Joint prescribing of omeprazolum with atazanaviry is not recommended.
Joint prescribing of omeprazolum (40 mg once a day) and an atazanavira of 300 mg / ritonavira led 100 mg at healthy volunteers to 75% to decrease in exposure of an atazanavir. Increase in a dose of an atazanavir up to 400 mg did not compensate influence of omeprazolum on exposure of an atazanavir. Joint intake of omeprazolum (20 mg once a day) from atazanaviry 400 mg / ritonavirom 100 mg for healthy volunteers led to decrease in exposure of an atazanavir approximately for 30% in comparison with atazanaviry 300 mg / ritonavirom 100 mg once a day.
Digoxin
Joint treatment by omeprazolum (20 mg a day) and digoxin at healthy subjects increased bioavailability of digoxin by 10%. About toxicity of digoxin it is reported seldom. However it is necessary to show care when assigning high doses of omeprazolum at patients of advanced age. It is necessary to strengthen therapeutic medicinal monitoring.
Klopidogrel
In cross clinical trials klopidogret (a load dose of 300 mg from the subsequent 75 mg/days) odinochno and together with omeprazolum use (80 mg at the same time as klopidogret) were accepted within 5 days. Exposure of an active metabolite of a klopidogrel was reduced by 46% (day 1) and 42% (day 5) at joint purpose of a klopidogrel and omeprazolum. The average inhibition of aggregation of thrombocytes (IPA) was reduced by 47% (24 hours) and 30% (day 5) at joint purpose of a klopidogrel and omeprazolum. In other research it was shown that reception of a klopidogrel and omeprazolum at different times does not interfere with their interaction which can be caused by inhibiting effect of omeprazolum on CYP2C19. In observation and clinical trials contradictory data on clinical manifestations of this FK/FD (pharmacokinetics/pharmacodynamics) of interaction were registered during the main cardiovascular events.
Other active agents
Absorption of a pozakonazol, erlotinib, ketokonazol and itrakonazol considerably decreases and, thus, clinical performance can be broken. For a pozakonazol and an erlotinib it is necessary to avoid simultaneous use.
Active agents, metaboliziruyemy CYP2C19
Omeprazol is moderate CYP2C19 inhibitor, the main enzyme metabolizing omeprazolum. Thus, metabolism of the accompanying active agents, also metaboliziruyemy CYP2C19, can be lowered, and system exposure of these substances can increase. Examples of such drugs are R-warfarin and other antagonists of vitamin K, tsilostazol, diazepam and Phenytoinum.
Tsilostazol
Omeprazol accepted in doses of 40 mg at healthy subjects in a cross research increased Cmax and AUC for a tsilostazol by 18% and 26% respectively, and one of its active metabolites for 29% and 69% respectively.
Phenytoinum
Within the first two weeks after an initiation of treatment is recommended by omeprazolum monitoring of concentration of Phenytoinum of plasma and if Phenytoinum dose adjustment was carried out, after the end of treatment the monitoring and further dose adjustment have to be carried out by omeprazolum.
Sakvinavir
Combined use of omeprazolum and a sakvinavira/ritonavir led to increase in level in plasma approximately up to 70% for the sakvinavir connected with good tolerance at patients with HIV infection.
Takrolimus
Pri combined use of omeprazolum increase in serumal levels of a takrolimus was observed. The strengthened monitoring of concentration of a takrolimus and also function of kidneys (clearance of creatinine) has to be executed, in need of a dosage of a takrolimus are adjusted.
Effect of other active agents on omeprazolum pharmacokinetics
CYP2C19 and/or SUR3A4 Inhibitors
As omeprazolum is metabolized by CYP2C19 and CYP3A4, the active agents known as CYP2C19 or SUR3A4 inhibitors (such as klaritromitsin and vorikonazol), can lead to increase in serumal levels of omeprazolum due to reduction of speed of his metabolism. Combined use of a vorikonazol brought to more than to omeprazolum exposure doubling. If high doses of omeprazolum are well transferred, omeprazolum dose adjustment usually is not required. However dose adjustment should be considered at patients with a heavy liver failure and at long-term treatment.
Inductors of isoenzymes of CYP2C19 and/or SUR3A4
the Active agents known as the inductors CYP2C19 or CYP3A4, or both (such as rifampicin and St. John's wort), can lead to decrease in level of omeprazolum in blood serum due to increase in speed of metabolism.
Special instructions
Before the beginning and after the end of treatment the endoscopic control for an exception of a malignant new growth as drug treatment omeprazolum can disguise symptomatology is obligatory and delay the correct diagnostics.
Pregnancy
Results of three perspective epidemiological researches (more than 1000 confirmed results) do not show negative consequences of omeprazolum on pregnancy or on health of the fruit / newborn. Omeprazol it is possible to apply during pregnancy.
The lactation
Omeprazol in breast milk, does not influence the child, at use of therapeutic doses.
Use for patients from the liver broken by function
Omeprazol is metabolized in a liver and at patients from the liver broken by function the elimination half-life of drug is extended therefore the drug dosage at such patients has to be reduced.
Special groups
the Liver failure
Metabolism of omeprazolum at patients from dysfunction of a liver is broken that leads to increase in AUC. Omeprazolum does not possess cumulation at reception once a day.
A renal failure
omeprazolum Pharmacokinetics, including system bioavailability and clearance rate, remain invariable with patients with reduced function of kidneys.
Adults
omeprazolum metabolism Speed decreases at elderly patients (75-79 years) a little.
Children's age
during treatment with the recommended doses for children aged from 1 year, similar concentration in plasma as at adults were received.
Features of influence on ability to run the vehicle or potentially dangerous mechanisms
Considering side effects of drug, it is necessary to be careful at control of transport or other types of activity demanding the increased speed of psychomotor reactions and concentration of attention.
Overdose
Symptoms: confusion of consciousness, drowsiness, a headache, disorders of vision, tachycardia, dryness in a mouth nausea, the increased sweating, and.
Treatment: gastric lavage, intake of activated carbon, symptomatic therapy.
A form of release and packing
On 10 capsules in planimetric bezjyacheykovy packing from aluminum foil. On 3 planimetric bezjyacheykovy packs together with the instruction for use in the state and Russian languages put in a pack from cardboard. On 5 packs put in a box of cardboard.
To Store storage conditions in the dry place protected from light at a temperature not above 25 °C.
To store out of children's reach!
3 years
not to apply a period of storage after an expiration date.
Prescription status
According to the prescription
Dr. of Reddi's Laboratoris Limited Producer, India
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products
Representative office Dr. of Reddi's Laboratoris Limited in Republic of Kazakhstan, 050057 Almaty, 22nd Line St., 45,
a post office box 7, ph. 8 (701) 7633805, fax: 8 (727) 3941294
To develop
of OMEZ® medicine
the Trade name
of Omez®
the International unlicensed
name Omeprazolum Dosage Form
of the Capsule kishechnorastvorimy 20 mg
One capsule contains Structure:
active agent - omeprazolum of 20 mg,
excipients: Mannitolum, lactose, sodium lauryl sulfate, dinatrium hydrophosphate anhydrous, sucrose, sucrose (25/30), gipromelloza (6cps)
main cover: gipromelloza (6 cps)
kishechnorastvorimy cover: methacrylic acid a kopolimer (type C) (eudragit L 100-55), sodium hydroxide, a macrogoal 6000, talc purified the titan E171 dioxide
structure of the capsule:
body: gelatin, methylparaben, propylparaben, sodium lauryl sulfate, E122 carmoisin,
lid: gelatin, methylparaben, propylparaben, sodium lauryl sulfate, karmoizin E122
the Description
Solid gelatin capsules No. 2 with the colourless transparent body and a transparent pink lid, with the inscription OMEZ on the body and a lid with the double blocking system. Contents of capsules – spherical pellets of white or almost white color.
Pharmacotherapeutic group
Antiulcerous drugs and drugs for treatment of a gastroegzofagealny reflux. Inhibitors of the protonew pump. Omeprazolum.
The ATX A02BC01 code
the Pharmacological
Pharmacokinetics Omeprazolum properties is put into acid resisting pellets which are dissolved only in intestines. At intake it is quickly absorbed from digestive tract. Meal does not affect bioavailability of drug. At reception of 20-40 mg of omeprazolum the bioavailability is about 40%, after repeated reception increases bioavailability about 60%. The peak of concentration of omeprazolum in blood plasma is reached within 0.5 - 3.5 hours. Communication with proteins of plasma - about 97%. Omeprazolum is metabolized in a liver. Metabolism is carried out through the system of P450 cytochrome (CYP2C19 isoform - S-Mephenytoinum a hydroxylase). Concentration of omeprazolum in plasma does not correlate with degree and duration of suppression of secretion of Acidum hydrochloricum (t½ - 0.5-1 hour, and the anti-secretory effect lasts up to 2-3 days).
The most part of drug of nearly 80% is removed by kidneys in the form of metabolites. Elimination half-life makes 0.5-1 hour.
The pharmacodynamics
Omeprazolum belongs to benzimidazolny derivatives. In acidic environment of covering cells of a stomach omeprazolum turns into an active metabolite - sulphenamide which it is irreversible contacts one of the N+,k cysteic groups +-ATF-azy and inhibits activity of enzyme (proton pomp). Thus, omeprazolum interferes with process of a kislotoobrazovaniye at the level of a final stage of synthesis of hydrochloric acid that leads to decrease in level of basal and stimulated secretion, irrespective of the irritant nature.
The inhibition of activity of enzyme has dose-dependent character.
Indications
Adults
- treatment of an ulcer of a duodenum, prevention of a recurrence of ulcers of a duodenum
- treatment of stomach ulcer, prevention of a recurrence of stomach ulcer
- in combination with the corresponding antibiotics, Helicobacter Pylori (H.P) eradication in a peptic ulcer
- treatment connected with intake of non-steroidal anti-inflammatory drugs (NPVP) - associated from gastropathies of a stomach and duodenum, prevention the gastropathies of a stomach and a duodenum connected with reception NPVP-assotsiirovannoy at patients with risk
- treatment a reflux esophagitis, long-term treatment of patients about a reflux esophagitis
- symptomatic treatment of a gastroesophagal reflux
- treatment of a syndrome
of Zollingera-Ellison Primeneniye in pediatrics
Children are more senior than 1 year and ≥ 10 kg
- treatment a reflux esophagitis
- symptomatic treatment of heartburn and an eructation acid in gastroesophagal a reflux
the Children's age is more senior than 4 years and teenage age
- in a combination with antibiotics in treatment of a peptic ulcer of a stomach and the duodenum caused by H. Pylori
the Route of administration and doses
Adult
the Recommended Omez's dose for treatment of a peptic ulcer of a duodenum – 20 mg (on 1 capsule) once a day within 4 weeks. Treatment can be prolonged for 4 weeks. For patients with a slabochuvstvitelny ulcer of a duodenum the use of Омез® 40 mg once a day, a course of treatment within 4 weeks is recommended.
The recommended Omez's dose for treatment of a peptic ulcer of a stomach – on 20 mg, two once a day (40 mg/days) within 4-8 weeks.
The recommended Omez's dose for a short course of treatment of a gastroesophageal reflux disease (GERD), not erosive esophagitis – 20 mg (on 1 capsule) daily once a day within 4 weeks.
The recommended Omez's dose for prevention of a recurrence of stomach ulcer - 20 mg once a day within 4 weeks. If necessary the dose of the drug Omez® can be increased up to 40 mg once a day, within 4 weeks.
For patients with an erosive esophagitis and with the complicated Omez GERD forms it is appointed in a dose of 20 mg (on 1 capsule) within 4 – 8 weeks.
In case of insufficient effectiveness of the above-stated therapy at patients with the I-II stages of GERD about 20 mg two times a day, within 4 - 8 weeks are recommended to accept, at III-IV stages of GERD recommend to accept on 40 mg once a day, within 4 - 8 weeks.
At more long-term treatment the dose of 20 mg at reception is appointed once a day, in the morning.
The recommended initial dose of Omez for the patients suffering from Zollingera-Ellison's syndrome – 60 mg. If necessary the dose is increased to 120 mg a day, dividing into 2 receptions.
Helicobacter pylori – the associated stomach diseases as a part of complex therapy: the recommended dose of 20 mg 2 times a day (it is strict in 12 hours, in the morning and in the evening) in a combination with antimicrobic therapy within 7 - 14 days.
- Omez of 20 mg + klaritromitsin 500 mg + amoxicillin of 1000 mg, two times a day within one week, or
- Omez of 20 mg + klaritromitsin 250 mg (or 500 mg) + metronidazole of 400 mg (either 500 mg or Tinidazolum of 500 mg), each drug two times a day within one week, or
- Omez of 40 mg once a day with amoxicillin of 500 mg and metronidazole of 400 mg (either 500 mg or Tinidazolum of 500 mg), both three times a day within one week.
The digestive tract erosive cankers connected with intake of non-steroidal anti-inflammatory drugs - the recommended dose - 20 mg two times (40 mg) a day within 4-8 weeks. For the purpose of prevention of appearance of new ulcers at use of non-steroidal anti-inflammatory drugs the long-term use of drug on 20 mg once a day, is recommended in the morning.
The digestive tract erosive cankers connected with intake of non-steroidal anti-inflammatory drugs - the recommended dose - 20 mg two times a day, within 4 - 8 weeks.
The recommended Omez's dose for prevention connected with intake of non-steroidal anti-inflammatory drugs - the associated stomach ulcer and a duodenum at patients of risk group (возраст>, 60, ulcer of stomach and duodenum in the anamnesis, earlier had gastrointestinal bleeding), the recommended dose - 20 mg once a day.
The recommended Omez's dose for treatment of a reflux esophagitis - 20 mg makes once a day, within 4 weeks. For patients with a heavy esophagitis the recommended Omez's dose of 40 mg once a day, within 8 weeks.
The recommended Omez's dose for treatment of a symptomatic gastroesophageal reflux - 20 mg once a day, within 4 weeks.
The recommended initial dose of Omez for the patients suffering from Zollingera-Ellison's syndrome - 60 mg once a day. If necessary the dose is increased to 120 mg a day, dividing into 2 receptions if the dose of drug exceeds 80 mg a day, it has to be divided and accepted two times a day.
Drug is recommended to be used before food, without damaging the capsule, to wash down with a small amount of liquid.
The children's age
Children is more senior than 1 year and & gt, 10 kg
Treatment of a reflux esophagitis
Symptomatic treatment of heartburn and regurgitation of acid in a gastroesophageal reflux disease, the recommended dose:
Age
Weight
Dosage
& gt, 1 year
of 10-20
kg 10 mg once a day. If necessary the dose can be increased up to 20 mg once a day
& gt, 2 years
& gt, 20
kg 20 mg once a day. If necessary the dose can be increased up to 40 mg
the Recommended Omez's dose for treatment a reflux esophagitis once a day - 20 mg, within 4 - 8 weeks.
The recommended Omez's dose at symptomatic treatment of heartburn and regurgitation of acid in a gastroesophageal reflux disease – 20 mg, within 2-4 weeks.
The children's age is more senior than 4 years and teenage age
the Recommended Omez's dose for treatment of a peptic ulcer of a stomach and a duodenum of pylori caused by N. – in courses of 1 - 2 week in a combination with antibacterial drugs:
Weight
the Dosage
of 15-30 kg
the Combination with two antibiotics: Omez of 10 mg, amoxicillin of 25 mg/kg of body weight and klaritromitsin 7.5 mg/kg of body weight are appointed in common two times a day within one week.
31-40 kg
the Combination with two antibiotics: Omez of 20 mg, amoxicillin of 750 mg and klaritromitsin 7.5 mg/kg of body weight are appointed in common two times a day within one week.
& gt, 40 kg
the Combination with two antibiotics: Omez of 20 mg, amoxicillin of 1 g and klaritromitsin 500 mg are appointed in common two times a day within one week.
The special group of the population
the Renal failure
with a renal failure dose adjustment is not required the Liver failure
At patients with a liver failure the patients can have a sufficient daily dose of 10-20 mg
Patients of advanced age (& gt, 65 years)
For patients of advanced age the dose adjustment is not required
the Route of administration
It is recommended to take the pill Omez in the morning, to swallow them entirely, washing down with a half of a glass of water.
For the patients having difficulties with swallowing and for children who can drink or swallow semi-fluid food
the Patients can dissolve it in a spoon of still water and if wish, to mix it with some fruit juice or apple puree. Patients have to be notified that dispersion has to be accepted immediately (or within 30 minutes), it should be mixed only just before the use and to wash down with a half of a glass of water. NOT to USE milk or sparkling water. Kishechnorastvorimy granules should not be chewed.
Side effects
- thorax pains, tachycardia or bradycardia, heartbeat, hypertensia, peripheral hypostases
- lack of appetite, a food faddism, an atrophy mucous language, dry language, esophageal candidiasis, a meteorism, irritation of intestines
- moderate tranzitorny increase in activity of transaminases, pancreatitis
- a hyponatremia, a hypoglycemia, increase in body weight
- spasms, muscle weakness, joint pains, the lower extremity pains
- a depression, aggression, hallucinations, drowsiness or insomnia, confusion of consciousness, nervousness, a tremor, apathy, feeling of alarm, dizziness, paresthesias
- disorders of vision, sonitus, nasal bleeding, a sore throat
- hemorrhagic rash and/or petechias, inflammation, an itching, dryness of integuments, a hyperhidrosis, an alopecia
- urticaria, an itching, a Quincke's disease
- interstitial nephrite, infections of urinary tract, a proteinuria, a hamaturia, a glucosuria
- pain in testicles, a gynecomastia
Seldom
- a neutropenia, a leukopenia, an agranulocytosis, a leukocytosis, thrombocytopenia, anemia, including hemolytic anemia, a pancytopenia
- the hepatocellular, cholestatic or mixed hepatitis
Very seldom
- a Lyell's disease, Stephens-Johnson's syndrome, a multiformny erythema
- risk of developing spinal fractures, necks of a hip,
the Contraindication wrist
- hypersensitivity to any of drug components
- simultaneous use with nelfinaviry
Medicinal interactions
of the Possibility of interaction of omeprazolum with other medicines are limited. Omeprazolum is metabolized in a liver through a system P450 cytochrome therefore at simultaneous use with drugs which are metabolized in a liver omeprazolum can reduce their removal. Omeprazolum slows down excretion of diazepam, Phenytoinum and anticoagulants (warfarin). At combined use with anticoagulants the control of blood test, a prothrombin time is necessary, correction of a dose of omeprazolum is necessary.
Nelfinavir, atazanavir
Plasma levels of a nelfinavir and atazanavir decrease in case of their combined use with omeprazolum.
Joint prescribing of omeprazolum with nelfinaviry contraindicated (see the section Simultaneous use of omeprazolum (40 mg once a day) reduces average exposure of a nelfinavir by 40%, and average exposure pharmacological of an active metabolite of M8 was reduced by 75-90%. Interaction can include CYP2C19 inhibition also.
Joint prescribing of omeprazolum with atazanaviry is not recommended.
Joint prescribing of omeprazolum (40 mg once a day) and an atazanavira of 300 mg / ritonavira led 100 mg at healthy volunteers to 75% to decrease in exposure of an atazanavir. Increase in a dose of an atazanavir up to 400 mg did not compensate influence of omeprazolum on exposure of an atazanavir. Joint intake of omeprazolum (20 mg once a day) from atazanaviry 400 mg / ritonavirom 100 mg for healthy volunteers led to decrease in exposure of an atazanavir approximately for 30% in comparison with atazanaviry 300 mg / ritonavirom 100 mg once a day.
Digoxin
Joint treatment by omeprazolum (20 mg a day) and digoxin at healthy subjects increased bioavailability of digoxin by 10%. About toxicity of digoxin it is reported seldom. However it is necessary to show care when assigning high doses of omeprazolum at patients of advanced age. It is necessary to strengthen therapeutic medicinal monitoring.
Klopidogrel
In cross clinical trials klopidogret (a load dose of 300 mg from the subsequent 75 mg/days) odinochno and together with omeprazolum use (80 mg at the same time as klopidogret) were accepted within 5 days. Exposure of an active metabolite of a klopidogrel was reduced by 46% (day 1) and 42% (day 5) at joint purpose of a klopidogrel and omeprazolum. The average inhibition of aggregation of thrombocytes (IPA) was reduced by 47% (24 hours) and 30% (day 5) at joint purpose of a klopidogrel and omeprazolum. In other research it was shown that reception of a klopidogrel and omeprazolum at different times does not interfere with their interaction which can be caused by inhibiting effect of omeprazolum on CYP2C19. In observation and clinical trials contradictory data on clinical manifestations of this FK/FD (pharmacokinetics/pharmacodynamics) of interaction were registered during the main cardiovascular events.
Other active agents
Absorption of a pozakonazol, erlotinib, ketokonazol and itrakonazol considerably decreases and, thus, clinical performance can be broken. For a pozakonazol and an erlotinib it is necessary to avoid simultaneous use.
Active agents, metaboliziruyemy CYP2C19
Omeprazol is moderate CYP2C19 inhibitor, the main enzyme metabolizing omeprazolum. Thus, metabolism of the accompanying active agents, also metaboliziruyemy CYP2C19, can be lowered, and system exposure of these substances can increase. Examples of such drugs are R-warfarin and other antagonists of vitamin K, tsilostazol, diazepam and Phenytoinum.
Tsilostazol
Omeprazol accepted in doses of 40 mg at healthy subjects in a cross research increased Cmax and AUC for a tsilostazol by 18% and 26% respectively, and one of its active metabolites for 29% and 69% respectively.
Phenytoinum
Within the first two weeks after an initiation of treatment is recommended by omeprazolum monitoring of concentration of Phenytoinum of plasma and if Phenytoinum dose adjustment was carried out, after the end of treatment the monitoring and further dose adjustment have to be carried out by omeprazolum.
Sakvinavir
Combined use of omeprazolum and a sakvinavira/ritonavir led to increase in level in plasma approximately up to 70% for the sakvinavir connected with good tolerance at patients with HIV infection.
Takrolimus
Pri combined use of omeprazolum increase in serumal levels of a takrolimus was observed. The strengthened monitoring of concentration of a takrolimus and also function of kidneys (clearance of creatinine) has to be executed, in need of a dosage of a takrolimus are adjusted.
Effect of other active agents on omeprazolum pharmacokinetics
CYP2C19 and/or SUR3A4 Inhibitors
As omeprazolum is metabolized by CYP2C19 and CYP3A4, the active agents known as CYP2C19 or SUR3A4 inhibitors (such as klaritromitsin and vorikonazol), can lead to increase in serumal levels of omeprazolum due to reduction of speed of his metabolism. Combined use of a vorikonazol brought to more than to omeprazolum exposure doubling. If high doses of omeprazolum are well transferred, omeprazolum dose adjustment usually is not required. However dose adjustment should be considered at patients with a heavy liver failure and at long-term treatment.
Inductors of isoenzymes of CYP2C19 and/or SUR3A4
the Active agents known as the inductors CYP2C19 or CYP3A4, or both (such as rifampicin and St. John's wort), can lead to decrease in level of omeprazolum in blood serum due to increase in speed of metabolism.
Special instructions
Before the beginning and after the end of treatment the endoscopic control for an exception of a malignant new growth as drug treatment omeprazolum can disguise symptomatology is obligatory and delay the correct diagnostics.
Pregnancy
Results of three perspective epidemiological researches (more than 1000 confirmed results) do not show negative consequences of omeprazolum on pregnancy or on health of the fruit / newborn. Omeprazol it is possible to apply during pregnancy.
The lactation
Omeprazol in breast milk, does not influence the child, at use of therapeutic doses.
Use for patients from the liver broken by function
Omeprazol is metabolized in a liver and at patients from the liver broken by function the elimination half-life of drug is extended therefore the drug dosage at such patients has to be reduced.
Special groups
the Liver failure
Metabolism of omeprazolum at patients from dysfunction of a liver is broken that leads to increase in AUC. Omeprazolum does not possess cumulation at reception once a day.
A renal failure
omeprazolum Pharmacokinetics, including system bioavailability and clearance rate, remain invariable with patients with reduced function of kidneys.
Adults
omeprazolum metabolism Speed decreases at elderly patients (75-79 years) a little.
Children's age
during treatment with the recommended doses for children aged from 1 year, similar concentration in plasma as at adults were received.
Features of influence on ability to run the vehicle or potentially dangerous mechanisms
Considering side effects of drug, it is necessary to be careful at control of transport or other types of activity demanding the increased speed of psychomotor reactions and concentration of attention.
Overdose
Symptoms: confusion of consciousness, drowsiness, a headache, disorders of vision, tachycardia, dryness in a mouth nausea, the increased sweating, and.
Treatment: gastric lavage, intake of activated carbon, symptomatic therapy.
A form of release and packing
On 10 capsules in planimetric bezjyacheykovy packing from aluminum foil. On 3 planimetric bezjyacheykovy packs together with the instruction for use in the state and Russian languages put in a pack from cardboard. On 5 packs put in a box of cardboard.
To Store storage conditions in the dry place protected from light at a temperature not above 25 °C.
To store out of children's reach!
3 years
not to apply a period of storage after an expiration date.
Prescription status
According to the prescription
Dr. of Reddi's Laboratoris Limited Producer, India
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products
Representative office Dr. of Reddi's Laboratoris Limited in Republic of Kazakhstan, 050057 Almaty, 22nd Line St., 45,
a post office box 7, ph. 8 (701) 7633805, fax: 8 (727) 3941294
To develop