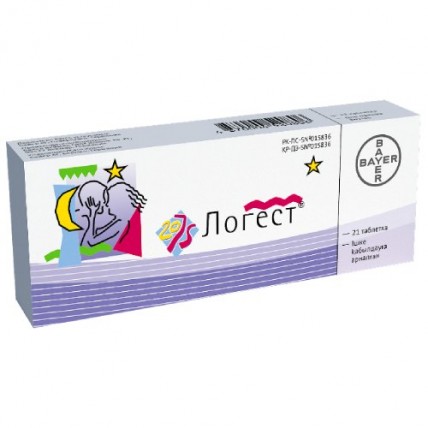
LOGEST® (Ethinylestradiol / Gestodene) 21 pills
- $33.00
LOGEST® are a hormonal contraceptive pill used to prevent pregnancy. It contains two active ingredients, Ethinylestradiol and Gestodene, which are synthetic forms of the female hormones estrogen and progestin, respectively.
LOGEST® (Ethinylestradiol / Gestodene) 21 pills work by preventing ovulation, or the release of an egg from the ovary, thereby making it difficult for fertilization to occur. Additionally, it also changes the cervical mucus and the lining of the uterus, making it harder for sperm to reach the egg or for a fertilized egg to implant in the uterus.
LOGEST® (Ethinylestradiol / Gestodene) 21 pills are taken orally once a day, with or without food, at the same time every day. The pills come in a pack of 21 tablets, and they should be taken continuously for 21 days, followed by a 7-day break, during which time a woman will have her menstrual period. After the 7-day break, a new pack of tablets is started, regardless of whether the menstrual period has stopped or not.
Each tablet of LOGEST® (Ethinylestradiol / Gestodene) 21 contains 0.03mg of ethinylestradiol and 0.075mg of gestodene. The recommended dosage is one tablet taken orally every day for 21 days, followed by a 7-day break.
Like all hormonal contraceptives, LOGEST® (Ethinylestradiol / Gestodene) 21 pills may cause side effects in some women, including headaches, nausea, breast tenderness, weight gain, mood changes, and irregular bleeding. Serious side effects are rare, but they may include blood clots, stroke, heart attack, and liver problems.
LOGEST® (Ethinylestradiol / Gestodene) 21 pills come with several warnings and precautions, including not being suitable for all women, particularly those with a history of blood clots, stroke, heart disease, liver disease, or breast cancer. Women who smoke are also at increased risk of serious side effects when taking hormonal contraceptives. It is important to discuss any medical conditions and medications with a healthcare provider before starting LOGEST® (Ethinylestradiol / Gestodene) 21 pills.
How Does LOGEST® (Ethinylestradiol / Gestodene) 21 pills Work?
LOGEST® (Ethinylestradiol / Gestodene) 21 pills work by preventing ovulation, or the release of an egg from the ovary, thereby making it difficult for fertilization to occur. Additionally, it also changes the cervical mucus and the lining of the uterus, making it harder for sperm to reach the egg or for a fertilized egg to implant in the uterus.
How is LOGEST® (Ethinylestradiol / Gestodene) 21 pills Taken?
LOGEST® (Ethinylestradiol / Gestodene) 21 pills are taken orally once a day, with or without food, at the same time every day. The pills come in a pack of 21 tablets, and they should be taken continuously for 21 days, followed by a 7-day break, during which time a woman will have her menstrual period. After the 7-day break, a new pack of tablets is started, regardless of whether the menstrual period has stopped or not.
What is the dosage of LOGEST® (Ethinylestradiol / Gestodene) 21 pills?
Each tablet of LOGEST® (Ethinylestradiol / Gestodene) 21 contains 0.03mg of ethinylestradiol and 0.075mg of gestodene. The recommended dosage is one tablet taken orally every day for 21 days, followed by a 7-day break.
What are the Side Effects of LOGEST® (Ethinylestradiol / Gestodene) 21 pills?
Like all hormonal contraceptives, LOGEST® (Ethinylestradiol / Gestodene) 21 pills may cause side effects in some women, including headaches, nausea, breast tenderness, weight gain, mood changes, and irregular bleeding. Serious side effects are rare, but they may include blood clots, stroke, heart attack, and liver problems.
What warnings does LOGEST® (Ethinylestradiol / Gestodene) 21 pills come with?
LOGEST® (Ethinylestradiol / Gestodene) 21 pills come with several warnings and precautions, including not being suitable for all women, particularly those with a history of blood clots, stroke, heart disease, liver disease, or breast cancer. Women who smoke are also at increased risk of serious side effects when taking hormonal contraceptives. It is important to discuss any medical conditions and medications with a healthcare provider before starting LOGEST® (Ethinylestradiol / Gestodene) 21 pills.