Glycine 100 mg sublingual (50 tablets)
- $5.00
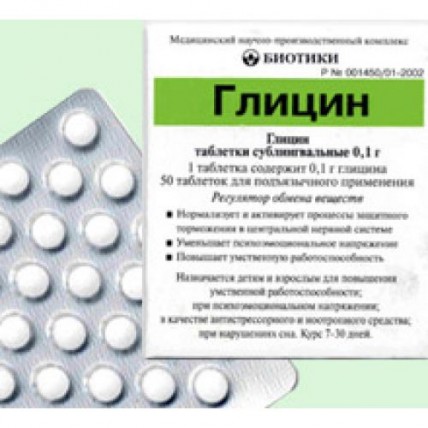
Glycine
Trade name: Glycine
International unlicensed name: glycine (aminoacetic acid).
Dosage form: hypoglossal tablets.
Structure.: glycine of 100 mg, excipients: methyl cellulose of water-soluble 1 mg, magnesium stearate of 1 mg.
Description: tablets of white color with elements of marbling, a ploskotsilindrichesky form with a facet.
Pharmacological group. Metabolic means.
ATH code: N06BX
Pharmacological properties. Glycine is the metabolism regulator, normalizes and activates processes of protective braking in the central nervous system, reduces psychoemotional pressure, increases intellectual working capacity. Glycine possesses glycine - and GAMK-ergichesky, alfa1-adrenoceptor blocking, antioxidant, anti-toxic action, regulates activity of glutamate (NMDA) receptors at the expense of what drug is capable:
to reduce psychoemotional pressure, aggression, conflictness, to increase social adaptation,
to improve mood,
to facilitate backfilling and to normalize a dream,
to increase intellectual working capacity,
to reduce vegeto-vascular disorders (including in a climacteric),
to reduce severity of brain disorders in an ischemic stroke and a craniocereberal injury,
to reduce toxic effect of alcohol and other medicines oppressing the central nervous system function.
Pharmacokinetics. Easily gets into the majority of biological liquids and body tissues, including into a brain, it is metabolized to water and carbon dioxide, its accumulation in fabrics does not happen.
Indications.
reduced intellectual working capacity.
stressful situations psychoemotional pressure (during examinations, situations conflict, etc.).
deviant forms of behavior of children and teenagers.
various functional and organic diseases of nervous system which are followed by hyperexcitability, emotional instability, decrease in intellectual working capacity and sleep disorder: neurosises, neurosis-like states and vegeto-vascular dystonia, consequences of neuroinfections and craniocereberal injury, perinatal and other forms of encephalopathies (including alcoholic genesis).
ischemic stroke.
Contraindications. Individual hypersensitivity to drug components.
Route of administration and doses. Glycine is applied podjyazychno or transbukkalno on 100 mg (in tablets or in the form of powder after crushing of a tablet). To almost healthy children, teenagers and adults at a psychoemotional tension, decrease in memory, attention, intellectual working capacity, a delay of intellectual development, at deviant forms of behavior glycine is appointed on 1 tablet 2-3 times a day within 14-30 days.
In the functional and organic lesions of nervous system which are followed by hyperexcitability, emotional lability and a sleep disorder, to children up to three years appoint 0.5 tablets (50 mg) to reception 2-3 times a day within 7-14 days, further on 50 mg 7-10 days once a day. A daily dose - 100-150 mg, course 2000-2600 mg. To children 3 years are more senior and the adult appoint on 1 tablet 2-3 times a day, a course of treatment of 7-14 days. The course of treatment can be increased up to 30 days, if necessary the course is repeated in 30 days.
In sleep disorders glycine is appointed in 20 minutes prior to a dream or just before a dream on 0.5-1 tablet (depending on age).
In an ischemic brain stroke: within the first 3-6 hours from development of a stroke appoint 1000 mg transbukkalno or podjyazychno with one teaspoon of water, further within 1-5 days on 1000 mg a day, then within the next 30 days 1-2 tablets 3 times a day. In narcology glycine is applied as the means increasing intellectual working capacity and reducing psychoemotional pressure during remission at the encephalopathy phenomena, organic lesions of the central and peripheral nervous system on 1 tablet 2-3 times a day within 14-30 days. If necessary courses repeat 4-6 times a year.
Side effect. Allergic reactions are possible.
Interaction with other medicines. Weakens severity of side effects of antipsychotic means (neuroleptics), anxiolytics, antidepressants, hypnotic drugs and anticonvulsants.
Release form. Tablets hypoglossal 100 mg.
Packaging. On 50 tablets in blister strip packaging in a pack cardboard together with the instruction for medical use.
Storage conditions. B list. In the dry place inaccessible for children protected from light, at a temperature not above 25o Page.
Expiration date. 3 years. Not to use after the expiry date specified on packing.
A release condition from drugstores. Without prescription of the doctor.
Trade name: Glycine
International unlicensed name: glycine (aminoacetic acid).
Dosage form: hypoglossal tablets.
Structure.: glycine of 100 mg, excipients: methyl cellulose of water-soluble 1 mg, magnesium stearate of 1 mg.
Description: tablets of white color with elements of marbling, a ploskotsilindrichesky form with a facet.
Pharmacological group. Metabolic means.
ATH code: N06BX
Pharmacological properties. Glycine is the metabolism regulator, normalizes and activates processes of protective braking in the central nervous system, reduces psychoemotional pressure, increases intellectual working capacity. Glycine possesses glycine - and GAMK-ergichesky, alfa1-adrenoceptor blocking, antioxidant, anti-toxic action, regulates activity of glutamate (NMDA) receptors at the expense of what drug is capable:
to reduce psychoemotional pressure, aggression, conflictness, to increase social adaptation,
to improve mood,
to facilitate backfilling and to normalize a dream,
to increase intellectual working capacity,
to reduce vegeto-vascular disorders (including in a climacteric),
to reduce severity of brain disorders in an ischemic stroke and a craniocereberal injury,
to reduce toxic effect of alcohol and other medicines oppressing the central nervous system function.
Pharmacokinetics. Easily gets into the majority of biological liquids and body tissues, including into a brain, it is metabolized to water and carbon dioxide, its accumulation in fabrics does not happen.
Indications.
reduced intellectual working capacity.
stressful situations psychoemotional pressure (during examinations, situations conflict, etc.).
deviant forms of behavior of children and teenagers.
various functional and organic diseases of nervous system which are followed by hyperexcitability, emotional instability, decrease in intellectual working capacity and sleep disorder: neurosises, neurosis-like states and vegeto-vascular dystonia, consequences of neuroinfections and craniocereberal injury, perinatal and other forms of encephalopathies (including alcoholic genesis).
ischemic stroke.
Contraindications. Individual hypersensitivity to drug components.
Route of administration and doses. Glycine is applied podjyazychno or transbukkalno on 100 mg (in tablets or in the form of powder after crushing of a tablet). To almost healthy children, teenagers and adults at a psychoemotional tension, decrease in memory, attention, intellectual working capacity, a delay of intellectual development, at deviant forms of behavior glycine is appointed on 1 tablet 2-3 times a day within 14-30 days.
In the functional and organic lesions of nervous system which are followed by hyperexcitability, emotional lability and a sleep disorder, to children up to three years appoint 0.5 tablets (50 mg) to reception 2-3 times a day within 7-14 days, further on 50 mg 7-10 days once a day. A daily dose - 100-150 mg, course 2000-2600 mg. To children 3 years are more senior and the adult appoint on 1 tablet 2-3 times a day, a course of treatment of 7-14 days. The course of treatment can be increased up to 30 days, if necessary the course is repeated in 30 days.
In sleep disorders glycine is appointed in 20 minutes prior to a dream or just before a dream on 0.5-1 tablet (depending on age).
In an ischemic brain stroke: within the first 3-6 hours from development of a stroke appoint 1000 mg transbukkalno or podjyazychno with one teaspoon of water, further within 1-5 days on 1000 mg a day, then within the next 30 days 1-2 tablets 3 times a day. In narcology glycine is applied as the means increasing intellectual working capacity and reducing psychoemotional pressure during remission at the encephalopathy phenomena, organic lesions of the central and peripheral nervous system on 1 tablet 2-3 times a day within 14-30 days. If necessary courses repeat 4-6 times a year.
Side effect. Allergic reactions are possible.
Interaction with other medicines. Weakens severity of side effects of antipsychotic means (neuroleptics), anxiolytics, antidepressants, hypnotic drugs and anticonvulsants.
Release form. Tablets hypoglossal 100 mg.
Packaging. On 50 tablets in blister strip packaging in a pack cardboard together with the instruction for medical use.
Storage conditions. B list. In the dry place inaccessible for children protected from light, at a temperature not above 25o Page.
Expiration date. 3 years. Not to use after the expiry date specified on packing.
A release condition from drugstores. Without prescription of the doctor.