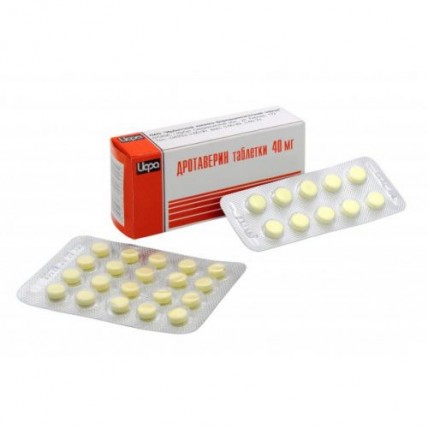
Drotaverina hydrochloride 40 mg (50 tablets)
- $3.10
The instruction for medical use
of medicine
Drotaverinum
the Trade name
Drotaverinum
the International unlicensed
name Drotaverinum Dosage Form
of the Tablet of 0.04 g
Structure
One tablet contains
active agent - Drotaverinum a hydrochloride
(in terms of 100% substance) – 0.04 g,
excipients: lactose monohydrate (sugar milk), potato starch, povidone (polyvinylpirrolidone low-molecular medical), magnesium stearate, talc.
The description
of the Tablet of color, yellow with a greenish shade, ploskotsilindrichesky with a facet
Pharmacotherapeutic group
Drugs for treatment of functional disorders of intestines.
Papaverine and its derivatives. Drotaverinum.
The ATX A03AD02 code
Pharmacological action
Pharmacokinetics
At oral administration absorption - high, the semi-absorption period - 12 min. Bioavailability - 100%. It is evenly distributed in fabrics, gets into smooth muscle cells. Time of achievement of the maximum concentration (TCmax) - 2 h. Communication with proteins of blood plasma - 95-98%. A half-life period - 2.4 h. It is generally removed by kidneys, to a lesser extent - with bile. Does not get through a blood-brain barrier (GEB).
The pharmacodynamics
Antispasmodic from group of derivatives of isoquinoline, makes expressed the weakening impact on smooth muscles of internals and vessels.
The mechanism of action is connected with inhibition of enzyme (FDE IV) phosphodiesterase IV. The inhibition of FDE IV leads to the termination of destruction of tsAMF and its accumulation in a smooth muscle cell. On chemical structure and pharmacological properties it is close to a papaverine, but possesses stronger and long action. Reduces a tone and physical activity of smooth muscles of internals, expands blood vessels.
In cells of smooth muscles of a myocardium and vessels, hydrolysis of tsAMF happens generally with the participation of FDE III isoenzyme that explains sharp selectivity of effect of Drotaverinum as spasmolysant in the absence of the significant therapeutic action on a cardiovascular system and development of the serious cardiovascular undesirable phenomena.
Indications
- spasms of smooth muscles in diseases of biliary tract: a cholecystolithiasis, a cholangiolithiasis, cholecystitis, a pericholecystitis, a cholangitis, a papillitis
- spasms of smooth muscles of mochevyvovdyashchy ways: a nephrolithiasis, an ureterolithiasis, a pyelitis, cystitis, bladder tenesmus
as auxiliary therapy:
- at spasms of smooth muscles of digestive tract: a peptic ulcer of a stomach and duodenum, gastritis, spasms of the cardia and the gatekeeper, enteritis, colitis, a spastic colitis with a constipation and a meteorism at a syndrome of the angry intestines
- in tension headaches
- in gynecologic diseases: a dysmenorrhea (painful periods)
the Route of administration and doses
the Adult appoint inside for 0.04-0.08 (1-2 tablets) 2-3 times a day. To children 12 years drug are more senior appoint 0.04 g (1 tablet) 1-3 times a day.
Side effects
Are possible
– dizziness, a headache, insomnia
- heartbeat, arterial hypotension
- feeling of heat, perspiration
- nausea, a constipation
- allergic reactions: infrequently Quincke's disease, a small tortoiseshell, rash,
naggers Protivopokazaniya
- hypersensitivity to Drotaverinum or to any excipient of drug
- a heavy renal or liver failure
- heavy heart failure (syndrome of low warm emission), AV blockade of the II-III degree
- the lactation period
- deficiency of lactose
- a galactosemia
- a syndrome of the broken absorption of a glucose/galactose
- children's age up to 12 years
Medicinal interactions
At simultaneous use Drotaverinum can weaken protivoparkinsonichesky effect of a levodopa.
At simultaneous use strengthens action of a papaverine, Bendazolum and other spasmolysants (including m-holinoblokatorov).
At simultaneous use with Drotaverinum of tricyclic antidepressants, quinidine and procaineamide their hypotensive action amplifies.
Special instructions
With care appoint drug to patients with the profound atherosclerosis of coronary arteries, prostate adenoma, glaucoma.
It is necessary to be careful when assigning to patients with pathology of coronary vessels and a benign hyperplasia of a prostate.
Pregnancy
Drotaverinum does not possess teratogenic and embriotoksichesky action. However use of drug is recommended only after careful weighing of a ratio advantage risk at mother and a fruit.
Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
Considering side effects, it is necessary to be careful at control of motor transport and potentially dangerous mechanisms.
Overdose
Symptoms: in high doses breaks atrioventricular conductivity, reduces excitability of a cardiac muscle, can cause cardiac arrest and paralysis of a respiratory center.
Treatment: drug withdrawal, the symptomatic therapy directed to elimination of the arisen disturbances. Specific antidote does not exist.
A form of release and packing
On 10 tablets in blister strip packaging from a film of the polyvinylchloride and printing aluminum foil varnished.
2 or 5 blister strip packagings with the instruction for medical use in the state and Russian languages place in a pack from cardboard.
To Store storage conditions in the dry, protected from light place at a temperature not over 25 ºС.
To store out of children's reach!
4 years
not to apply a period of storage after an expiration date.
Prescription status
Without prescription
JSC Irbit Chemical Pharmaceutical Plant Producer 623856,
Sverdlovsk Region, Irbit, Kirov St., 172 Ph. / fax (34355) 3-60-90
The name and the country of the owner of the registration certificate
of JSC Irbit Chemical and Pharmaceutical Plant, Russia
the Address of the organization accepting claims from consumers on quality of products (goods) of JSC Irbit Chemical Pharmaceutical Plant 623856, Sverdlovsk Region, Irbit, Kirov St., 172tel/fax (34355) 3-60-90Адрес e-mail: info@ihfz.ru
of medicine
Drotaverinum
the Trade name
Drotaverinum
the International unlicensed
name Drotaverinum Dosage Form
of the Tablet of 0.04 g
Structure
One tablet contains
active agent - Drotaverinum a hydrochloride
(in terms of 100% substance) – 0.04 g,
excipients: lactose monohydrate (sugar milk), potato starch, povidone (polyvinylpirrolidone low-molecular medical), magnesium stearate, talc.
The description
of the Tablet of color, yellow with a greenish shade, ploskotsilindrichesky with a facet
Pharmacotherapeutic group
Drugs for treatment of functional disorders of intestines.
Papaverine and its derivatives. Drotaverinum.
The ATX A03AD02 code
Pharmacological action
Pharmacokinetics
At oral administration absorption - high, the semi-absorption period - 12 min. Bioavailability - 100%. It is evenly distributed in fabrics, gets into smooth muscle cells. Time of achievement of the maximum concentration (TCmax) - 2 h. Communication with proteins of blood plasma - 95-98%. A half-life period - 2.4 h. It is generally removed by kidneys, to a lesser extent - with bile. Does not get through a blood-brain barrier (GEB).
The pharmacodynamics
Antispasmodic from group of derivatives of isoquinoline, makes expressed the weakening impact on smooth muscles of internals and vessels.
The mechanism of action is connected with inhibition of enzyme (FDE IV) phosphodiesterase IV. The inhibition of FDE IV leads to the termination of destruction of tsAMF and its accumulation in a smooth muscle cell. On chemical structure and pharmacological properties it is close to a papaverine, but possesses stronger and long action. Reduces a tone and physical activity of smooth muscles of internals, expands blood vessels.
In cells of smooth muscles of a myocardium and vessels, hydrolysis of tsAMF happens generally with the participation of FDE III isoenzyme that explains sharp selectivity of effect of Drotaverinum as spasmolysant in the absence of the significant therapeutic action on a cardiovascular system and development of the serious cardiovascular undesirable phenomena.
Indications
- spasms of smooth muscles in diseases of biliary tract: a cholecystolithiasis, a cholangiolithiasis, cholecystitis, a pericholecystitis, a cholangitis, a papillitis
- spasms of smooth muscles of mochevyvovdyashchy ways: a nephrolithiasis, an ureterolithiasis, a pyelitis, cystitis, bladder tenesmus
as auxiliary therapy:
- at spasms of smooth muscles of digestive tract: a peptic ulcer of a stomach and duodenum, gastritis, spasms of the cardia and the gatekeeper, enteritis, colitis, a spastic colitis with a constipation and a meteorism at a syndrome of the angry intestines
- in tension headaches
- in gynecologic diseases: a dysmenorrhea (painful periods)
the Route of administration and doses
the Adult appoint inside for 0.04-0.08 (1-2 tablets) 2-3 times a day. To children 12 years drug are more senior appoint 0.04 g (1 tablet) 1-3 times a day.
Side effects
Are possible
– dizziness, a headache, insomnia
- heartbeat, arterial hypotension
- feeling of heat, perspiration
- nausea, a constipation
- allergic reactions: infrequently Quincke's disease, a small tortoiseshell, rash,
naggers Protivopokazaniya
- hypersensitivity to Drotaverinum or to any excipient of drug
- a heavy renal or liver failure
- heavy heart failure (syndrome of low warm emission), AV blockade of the II-III degree
- the lactation period
- deficiency of lactose
- a galactosemia
- a syndrome of the broken absorption of a glucose/galactose
- children's age up to 12 years
Medicinal interactions
At simultaneous use Drotaverinum can weaken protivoparkinsonichesky effect of a levodopa.
At simultaneous use strengthens action of a papaverine, Bendazolum and other spasmolysants (including m-holinoblokatorov).
At simultaneous use with Drotaverinum of tricyclic antidepressants, quinidine and procaineamide their hypotensive action amplifies.
Special instructions
With care appoint drug to patients with the profound atherosclerosis of coronary arteries, prostate adenoma, glaucoma.
It is necessary to be careful when assigning to patients with pathology of coronary vessels and a benign hyperplasia of a prostate.
Pregnancy
Drotaverinum does not possess teratogenic and embriotoksichesky action. However use of drug is recommended only after careful weighing of a ratio advantage risk at mother and a fruit.
Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
Considering side effects, it is necessary to be careful at control of motor transport and potentially dangerous mechanisms.
Overdose
Symptoms: in high doses breaks atrioventricular conductivity, reduces excitability of a cardiac muscle, can cause cardiac arrest and paralysis of a respiratory center.
Treatment: drug withdrawal, the symptomatic therapy directed to elimination of the arisen disturbances. Specific antidote does not exist.
A form of release and packing
On 10 tablets in blister strip packaging from a film of the polyvinylchloride and printing aluminum foil varnished.
2 or 5 blister strip packagings with the instruction for medical use in the state and Russian languages place in a pack from cardboard.
To Store storage conditions in the dry, protected from light place at a temperature not over 25 ºС.
To store out of children's reach!
4 years
not to apply a period of storage after an expiration date.
Prescription status
Without prescription
JSC Irbit Chemical Pharmaceutical Plant Producer 623856,
Sverdlovsk Region, Irbit, Kirov St., 172 Ph. / fax (34355) 3-60-90
The name and the country of the owner of the registration certificate
of JSC Irbit Chemical and Pharmaceutical Plant, Russia
the Address of the organization accepting claims from consumers on quality of products (goods) of JSC Irbit Chemical Pharmaceutical Plant 623856, Sverdlovsk Region, Irbit, Kirov St., 172tel/fax (34355) 3-60-90Адрес e-mail: info@ihfz.ru