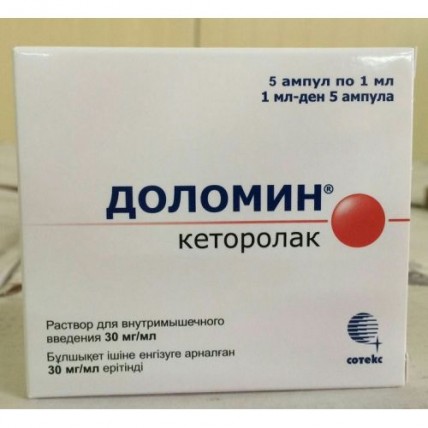
Dolomin 30 mg / ml 1ml 5's solution for intramuscular administration
- $18.20
The instruction for medical use of Dolomin® medicine the Trade name of Dolomin® the International unlicensed name Ketorolak Lekarstvennaya a form Solution for intramuscular introduction of 30 mg/ml Structure One ampoule (1 ml) contains active agent - a ketorolak trometamol 30 mg, excipients: ethanol (alcohol of 95% in terms of 100%), sodium chloride, 1 M solution of sodium of hydroxide or 1 M chlorohydrogen acid, water for injections the Description Transparent liquid of light yellow color Pharmacotherapeutic group Anti-inflammatory and antirheumatic drugs. Non-steroidal anti-inflammatory drugs. Acetic acid derivatives. Ketorolak the ATX M01AB15 Code the Pharmacological Pharmacokinetics Maximum Concentration properties (Cmax) after intramuscular introduction of 30 mg – 1.74-3.1 mkg/ml, 60 mg – 3.23-5.77 mkg/ml. Time of achievement of the maximum concentration (TCmax) – 15-70 min. Communication with proteins of plasma – 99%. Time of achievement of equilibrium concentration (Css) at parenteral administration – 24 h when assigning 4 times a day (above subtherapeutic) also is at intramuscular introduction 15 mg – 0.5-1 mkg/ml, 30 mg – 1-2.5 mkg/ml. Distribution volume – 0.15-0.3 l/kg. Gets into breast milk. More than 50% of the entered dose are metabolized in a liver with formation pharmacological of inactive metabolites. The main metabolites are glucuronides which are removed by kidneys and r-hydroksiketorolak. 6% are brought to 90% by kidneys, – through intestines. Elimination half-life (T1/2) at patients with normal function of kidneys – on average 5.3 h (3.5-9.2 h after intramuscular introduction of 30 mg). T1/2 increases at elderly patients and is shortened at young people. Function of a liver does not influence T1/2. At patients with a renal failure at concentration of creatinine in plasma of 19-50 mg/l (168 - 442 µmol/l) T1/2 – 10.3-10.8 h, in more profound renal failure – more than 13.6 h. The general clearance is at intramuscular introduction 30 mg – 0.023 l/h/kg (elderly patients have 0.019 l/h/kg), at patients with a renal failure at concentration of creatinine in plasma of 19-50 mg/l at intramuscular introduction of 30 mg – 0.015 l/h/kg. It is not removed by a hemodialysis. Pharmacodynamics Non-steroidal anti-inflammatory drug (NPVP). Has the significant analgeziruyushchy effect, possesses also anti-inflammatory and moderate febrifugal actions. The mechanism of action is connected with non-selective oppression of activity of cyclooxygenase-1 and cyclooxygenase-2, catalyzing formation of prostaglandins from arachidonic acid which play an important role in pathogenesis of pain, inflammation and fever. After intramuscular introduction the beginning of analgeziruyushchy action is noted in 0.5 h, the maximum effect is reached in 1-2 h. Indications the Pain syndrome of strong and moderate expressiveness - injuries, dislocations, stretchings - a toothache - pains in the postoperative period - oncological diseases - myalgias, arthralgias, neuralgia, sciatica, rheumatic diseases the Route of administration and doses Intramusculary (in oil). Patients are from 18 to 64 years old with the body weight exceeding 50 kg in oil for one introduction enter no more than 60 mg (including an oral dose), usually on 30 mg each 6 h. To adult patients with body weight less than 50 kg or with the chronic kidney disease (CKD) for 1 introduction in oil enter no more than 30 mg (including an oral dose). The maximum daily dose for introduction in oil for patients from 18 to 64 years with the body weight exceeding 50 kg makes 90 mg/days, for adult patients with body weight less than 50 kg or with HPN and also for elderly patients (65 years are more senior) of 60 mg a day. Duration of treatment should not exceed 5 days. Side effects Often – more than 3%, infrequently – 1-3%, are rare – less than 1% Often gastralgias, diarrhea a headache, dizziness, drowsiness hypostases (persons, shins, anklebones, fingers, a foot, increase in body weight) Infrequently stomatitis vomiting, feeling of overflow of a stomach, a meteorism, a constipation increase in arterial blood pressure burning or pain in the injection site the increased perspiration skin rash (including makulopapullezny rash), a purpura Seldom nausea, digestive tract erosive cankers (including with perforation and/or bleeding – abdominal pain, a spasm or burning in epigastric area, blood in Calais or a melena, vomiting with blood or as a coffee thick, heartburn, etc.) cholestatic jaundice, a hepatomegalia, hepatitis, acute pancreatitis a back pain with or without hamaturia and/or an azotemia, frequent urination, increase or decrease in volume of urine, hypostases of renal genesis nephrite, an acute renal failure, a gemolitiko-uraemic syndrome (hemolytic anemia, a renal failure, thrombocytopenia, a purpura) decrease in hearing, a ring in ears, a disorder of vision (including illegibility of visual perception) a bronchospasm or dispnoe, rhinitis, a fluid lungs, a laryngeal edema (asthma, breath difficulty) aseptic meningitis (fever, a severe headache, spasms, muscle tension of a neck and/or back), hyperactivity (change of mood, concern), hallucinations, a depression, psychosis a syncope anemia, an eosinophilia, a leukopenia bleeding from a postoperative wound, nasal bleeding, rectal bleeding exfoliative dermatitis (fever with a fever or without, reddening, consolidation or peeling of skin, swelling and/or morbidity of palatine tonsils), a small tortoiseshell, Stephens-Johnson's syndrome, a Lyell's disease an anaphylaxis or anaphylactoid reactions (discoloration of face skin, skin rash, a small tortoiseshell, an itching of skin, a tachypnea or dispnoe, swell a century, periorbital hypostasis, an asthma, the complicated breath, weight in a thorax, goose breathing) a paraglossa, Contraindication fever hypersensitivity to a ketorolak, other non-steroidal anti-inflammatory drugs or to one of excipients a 'aspirinovy' triad (a combination of bronchial asthma, the nose recuring a polypose and near-nasal bosoms and intolerance of acetylsalicylic acid and medicines of a pyrazolon row) a hypovolemia (irrespective of the reason which caused to it) digestive tract erosive cankers in an aggravation stage hypocoagulation (including hemophilia), bleedings or high risk of their development a heavy renal failure (creatinine of plasma is higher than 50 mg/l) a heavy liver failure the period of pregnancy and a lactation (gets into breast milk in insignificant quantities) children's age (up to 18 years – safety and efficiency are not established). With care Bronchial asthma, existence of the factors increasing toxicity concerning digestive tract: alcoholism, tobacco smoking, cholecystitis, chronic heart failure, an edematous syndrome, arterial hypertension, a renal failure (creatinine of plasma is lower than 50 mg/l), a cholestasia, active hepatitis, sepsis, a system lupus erythematosus, a concomitant use with other NPVP or advanced age (65 years are more senior). Medicinal interactions Joint appointment with paracetamol increases nephrotoxicity of a ketorolak. Reception with other NPVP, glucocorticosteroids, ethanol, corticotropin, drugs of calcium increases risk of an ulceration of a mucous membrane of digestive tract and development of gastrointestinal bleedings. Co-administration with anticoagulants – derivatives of coumarin and an indandion, heparin, trombolitika (alteplaza, streptokinase, an urokinase), antiagregant, cephalosporins, valproic acid and acetylsalicylic acid increases risk of developing bleedings. Reduces effect of hypotensive and diuretic medicines (reduces synthesis of prostaglandins in kidneys). Appointment together with a methotrexate raises gepato- and nephrotoxicity (their joint appointment is possible only during the using of low doses of the last and control of its concentration in plasma). Probenetsid reduces plasma clearance and volume of distribution of a ketorolak, increases its concentration in blood plasma and increases the period of its semi-removal. Against the background of use of a ketorolak the reduction of clearance of a methotrexate and lithium and strengthening of toxicity of these substances is possible. Co-administration with indirect anticoagulants, heparin, trombolitika, antiagregant, tsefoperazony, tsefotetany and pentoksifilliny increases risk of bleeding. When assigning with other nefrotoksichny medicines (including with gold drugs) risk of development of nephrotoxicity increases. The medicines blocking canalicular secretion reduce clearance of a ketorolak and increase its concentration in plasma. Increases effect of narcotic analgetics. Miyelotoksichny medicines strengthen manifestation of a gematotoksichnost of drug. At a concomitant use of a ketorolak with psychotropic drugs (fluoxetine, tiotiksen, to alprazola) at patients hallucinations were noted. Special instructions Before prescribing of drug it is necessary to find out a question of the previous allergy to drug or NPVP. Because of risk of development of allergic reactions the introduction of the first dose is carried out under careful observation of the doctor. Drug contains 100 mg of alcohol in 1 ml of solution therefore it can be dangerous to the persons having alcoholism, to patients with diseases of a liver and epilepsy. The hypovolemia increases risk of development of nephrotoxic side reactions. If necessary it is possible to appoint in a combination with narcotic analgetics. It is not recommended to apply as medicine to premedication, anesthesia maintenance. At joint reception with other NPVP the liquid delay, a decompensation of warm activity, increase in arterial blood pressure can be observed. Influence on aggregation of thrombocytes stops in 24-48 h. Not to use along with paracetamol more than 5 days. The patient with disturbance of fibrillation appoint only at constant control of number of thrombocytes, it is especially important for the postoperative patients demanding careful control of a hemostasis. Risk of development of medicinal complications increases when lengthening treatment (at patients with chronic pains) and increase in a dose of drug more than 40 mg/days. Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms. Due to the possible emergence of drowsiness and dizziness during use of a ketorolak, it is necessary to be careful during the driving of motor transport and occupation other potentially dangerous types of activity demanding the increased concentration of attention and speed of psychomotor reactions. Overdose Symptoms (at single introduction) an abdominal pain, nausea, vomiting, a digestive tract erosive canker, a renal failure, a metabolic acidosis. Treatment of symptomatic (maintenance of the vital functions of an organism). Dialysis is ineffective. A form of release and packing Solution for intramuscular introduction of 30 mg/ml. On 1 ml in ampoules of light-protective glass with a color ring of a break or with a color point and a notch. One, two or three color rings and/or the two-dimensional barcode, and/or alphanumeric coding or without additional color rings, the two-dimensional barcode, alphanumeric coding in addition apply on ampoules. On 5 ampoules place in blister strip packaging from a film of polyvinylchloride and aluminum foil or a film polymeric or without foil and without film. 1 or 2 blister strip packagings together with the instruction for medical use in the state and Russian languages in a pack from cardboard. To Store storage conditions in the place protected from light at a temperature from 15 to 25 ºС. To store out of children's reach! 4 years not to use a period of storage after the expiration date specified on packing. The Closed joint stock company Producer Farmfirma of Sotex, Russia 141345, the Moscow Region, the Sergiyevo-Posadsky municipal district, the Bereznyakovskoye rural settlement, the settlement of Belikovo, 11 the Name and the country of the owner of the registration certificate Closed joint stock company Farmfirma of Sotex, Russia the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products (goods) of EL company (EL company) LLP Republic of Kazakhstan, Almaty, Masangchi St., 98A, office 41
to Develop prescription status According to the prescription
to Develop prescription status According to the prescription