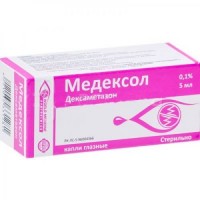
Diclofenac 0.1% / 5 ml eye drop
- $6.60
The instruction for use
of medicine for experts
Diclofenac
the Trade name
Diclofenac
the International unlicensed
name Sodium Diclofenac
the Dosage form
Eye drops, 0.1%
Structure
of 1 ml of solution contains
active agent – diclofenac of sodium of 1 mg,
excipients: EL cremophore, trometamol, a benzalkoniya chloride, dinatrium edetat a dihydrate, Mannitolum, the hydrochloric acid, water purified.
The description
Transparent liquid without mechanical particles
Pharmacotherapeutic group
Drugs for treatment of diseases of eyes.
Non-steroidal anti-inflammatory drugs (NPVP).
The code of automatic telephone exchange S01BC03
the Pharmacological
Pharmacokinetics At properties of instillation of eye drops Diclofenac is soaked up in a system blood stream. However the concentration of medicinal substance reached in blood is much lower than a limit of detection and has no clinical importance.
A pharmacodynamics
Diclofenac – the non-steroidal anti-inflammatory drug derivative of phenylacetic acid. Has the significant anti-inflammatory and analgeziruyushchy effect. The mechanism of action is connected with oppression of activity of cyclooxygenase (COG) – key enzyme of metabolism of arachidonic acid, being the predecessor of prostaglandins which play an important role in pathogenesis of inflammation and pain. The analgetic effect of Diclofenac is directly connected with its anti-inflammatory action which is a consequence of oppression of synthesis of prostaglandins in the inflamed fabrics.
Diclofenac reduces aggregation of thrombocytes. At topical administration in ophthalmology reduces hypostasis and pain at inflammatory processes.
Indications
- conjunctivitis (including seasonal allergic), a keratoconjunctivitis, a cornea erosion, posttraumatic inflammation of a conjunctiva and cornea
- treatment and prevention of posttraumatic inflammatory process at the getting and not getting eyeball wounds (as addition to local anti-infectious therapy)
- treatment and prevention of inflammatory processes after surgical interventions on an eyeball
- miosis inhibition during operation for a cataract
- prevention of cystous hypostasis of a makula after operations for a cataract
- a pain syndrome when carrying out excimer laser intervention
- photophobia after keratotomy
the Route of administration and doses
Before burying carefully wash up hands.
Drug intends for local superficial use, is not entered subkonjyuktivalno or into an anterior chamber of an eye.
For inhibition of an intraoperative miosis drug is dug in in a conjunctival sac within 2 hours with an interval of 30 minutes (only 4 times) before operation. After operation - on 1 drop of 3-5 times a day during time, necessary for treatment.
For prevention of cystous hypostasis of a makula drug is dug in on 1 drop of 3-4 times/days within 2 weeks after operation.
At other damages of eyes of various etiology about 1 drop of 3-4 times a day depending on severity of a disease is applied.
At more long-term treatment the detailed ophthalmologic inspection and exact diagnostics is necessary. The course of treatment can proceed from 1 to 2 weeks.
If you accept Diclofenac in combination with other eye drops, keep the minimum interval 15 minutes between burying.
If you forgot to use one dose, its instilliruyta as soon as possible. Do not dig in a double dose for compensation forgotten.
At use to avoid touch of a cover dropper of an eye or a zone around eyes for maintaining sterility of solution. In order to avoid any bacterial infection avoid contact of a cover dropper to something. Close a bottle after the use.
Side effects
- passing burning sensation and/or temporary loss of clearness of sight directly after burying
- an itching, reddening, a photosensitization, a dot keratitis
- urticaria, nausea, vomiting, a Quincke's disease of the person, fever, a fever.
Contraindications
- hypersensitivity to any component of drug
- hypersensitivity to acetylsalicylic acid and other non-steroidal anti-inflammatory drugs
- the III trimester of pregnancy
- children's age (up to 2 years).
Medicinal interactions
in the presence of clinical indications can be combined with the eye drops containing glucocorticosteroid means. Though system absorption is minimum, diclofenac of sodium can interact with other medicines. Increase in risk of a hyperpotassemia is promoted co-administration with potassium salts, kaliysberegayushchy diuretic, cyclosporine, Trimethoprimum. Other non-steroidal anti-inflammatory drugs, oral anticoagulants and heparin can increase risk of appearance of an ulcer and bleedings. Diclofenac of sodium can increase toxicity of lithium by means of decrease in its renal removal. Diuretics and inhibitors of enzyme of conversion of angiotensin II increase risk of a renal failure by oppression of synthesis of vasodilating prostaglandins.
If necessary co-administration of other medicines in the form of eye drops is possible, but the break between instillations has to be not less than 5 min. for prevention of washing away of active ingredients.
Special instructions
With care apply in an epithelial herpetic keratitis, in the diseases causing disturbances of processes of fibrillation (including hemophilia, lengthening of a bleeding time, tendency to bleedings).
The benzalkoniya chloride (preservative) which can be laid for surfaces of soft contact lenses is a part of drug therefore lenses need to be removed in 15 minutes prior to burying, their carrying is possible not earlier than 15 minutes after burying.
Use in pediatrics
Safety of use of eye drops Diclofenac at children is not established. Appointment is possible only in cases when the expected advantage of therapy exceeds risk of use of drug for this category of patients.
Pregnancy and a lactation
of Adequate and strictly controlled researches of safety of use at pregnancy and in the period of a lactation (breastfeeding) it is not carried out. Use of drug for this category of patients is possible only if the expected advantage of therapy for mother exceeds potential risk for a fruit or the baby.
Influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
does not influence.
Patients at whom after applique the sight clearness is temporarily lost are not recommended to drive or work with a difficult technique right after burying drug.
The overdose
At use of eye drops Diclofenac in the recommended doses
overdose is improbable. Now about cases of overdose of the drug Diclofenac it was not reported.
Overdose symptoms: strengthening of side effects of drug.
Treatment: to wash eyes a large number with warm water.
A form of release and packing
Eye drops, solution of 0.1% of diclofenac of sodium
On 5 ml in a white bottle from polyethylene with a stopper dropper and the screwing-up protective cap supplied with a safety ring.
1 bottle together with the instruction for use in a cardboard pack.
To Store storage conditions at a temperature from + 150C to + 250C in original packing.
To store out of children's reach!
A period of storage
3 years
do not use drug after the expiry date specified on packing.
After opening the bottle should be stored in the fridge within 28 days.
Prescription status
According to the prescription
of Proizvoditel Rompharm Company of S.R. L.,
Romania Eroilor St. 1A, Otopen.
ph. +40 21 208 9743, +40 21 208 97 42, fax: +40 21 266 49 38
of medicine for experts
Diclofenac
the Trade name
Diclofenac
the International unlicensed
name Sodium Diclofenac
the Dosage form
Eye drops, 0.1%
Structure
of 1 ml of solution contains
active agent – diclofenac of sodium of 1 mg,
excipients: EL cremophore, trometamol, a benzalkoniya chloride, dinatrium edetat a dihydrate, Mannitolum, the hydrochloric acid, water purified.
The description
Transparent liquid without mechanical particles
Pharmacotherapeutic group
Drugs for treatment of diseases of eyes.
Non-steroidal anti-inflammatory drugs (NPVP).
The code of automatic telephone exchange S01BC03
the Pharmacological
Pharmacokinetics At properties of instillation of eye drops Diclofenac is soaked up in a system blood stream. However the concentration of medicinal substance reached in blood is much lower than a limit of detection and has no clinical importance.
A pharmacodynamics
Diclofenac – the non-steroidal anti-inflammatory drug derivative of phenylacetic acid. Has the significant anti-inflammatory and analgeziruyushchy effect. The mechanism of action is connected with oppression of activity of cyclooxygenase (COG) – key enzyme of metabolism of arachidonic acid, being the predecessor of prostaglandins which play an important role in pathogenesis of inflammation and pain. The analgetic effect of Diclofenac is directly connected with its anti-inflammatory action which is a consequence of oppression of synthesis of prostaglandins in the inflamed fabrics.
Diclofenac reduces aggregation of thrombocytes. At topical administration in ophthalmology reduces hypostasis and pain at inflammatory processes.
Indications
- conjunctivitis (including seasonal allergic), a keratoconjunctivitis, a cornea erosion, posttraumatic inflammation of a conjunctiva and cornea
- treatment and prevention of posttraumatic inflammatory process at the getting and not getting eyeball wounds (as addition to local anti-infectious therapy)
- treatment and prevention of inflammatory processes after surgical interventions on an eyeball
- miosis inhibition during operation for a cataract
- prevention of cystous hypostasis of a makula after operations for a cataract
- a pain syndrome when carrying out excimer laser intervention
- photophobia after keratotomy
the Route of administration and doses
Before burying carefully wash up hands.
Drug intends for local superficial use, is not entered subkonjyuktivalno or into an anterior chamber of an eye.
For inhibition of an intraoperative miosis drug is dug in in a conjunctival sac within 2 hours with an interval of 30 minutes (only 4 times) before operation. After operation - on 1 drop of 3-5 times a day during time, necessary for treatment.
For prevention of cystous hypostasis of a makula drug is dug in on 1 drop of 3-4 times/days within 2 weeks after operation.
At other damages of eyes of various etiology about 1 drop of 3-4 times a day depending on severity of a disease is applied.
At more long-term treatment the detailed ophthalmologic inspection and exact diagnostics is necessary. The course of treatment can proceed from 1 to 2 weeks.
If you accept Diclofenac in combination with other eye drops, keep the minimum interval 15 minutes between burying.
If you forgot to use one dose, its instilliruyta as soon as possible. Do not dig in a double dose for compensation forgotten.
At use to avoid touch of a cover dropper of an eye or a zone around eyes for maintaining sterility of solution. In order to avoid any bacterial infection avoid contact of a cover dropper to something. Close a bottle after the use.
Side effects
- passing burning sensation and/or temporary loss of clearness of sight directly after burying
- an itching, reddening, a photosensitization, a dot keratitis
- urticaria, nausea, vomiting, a Quincke's disease of the person, fever, a fever.
Contraindications
- hypersensitivity to any component of drug
- hypersensitivity to acetylsalicylic acid and other non-steroidal anti-inflammatory drugs
- the III trimester of pregnancy
- children's age (up to 2 years).
Medicinal interactions
in the presence of clinical indications can be combined with the eye drops containing glucocorticosteroid means. Though system absorption is minimum, diclofenac of sodium can interact with other medicines. Increase in risk of a hyperpotassemia is promoted co-administration with potassium salts, kaliysberegayushchy diuretic, cyclosporine, Trimethoprimum. Other non-steroidal anti-inflammatory drugs, oral anticoagulants and heparin can increase risk of appearance of an ulcer and bleedings. Diclofenac of sodium can increase toxicity of lithium by means of decrease in its renal removal. Diuretics and inhibitors of enzyme of conversion of angiotensin II increase risk of a renal failure by oppression of synthesis of vasodilating prostaglandins.
If necessary co-administration of other medicines in the form of eye drops is possible, but the break between instillations has to be not less than 5 min. for prevention of washing away of active ingredients.
Special instructions
With care apply in an epithelial herpetic keratitis, in the diseases causing disturbances of processes of fibrillation (including hemophilia, lengthening of a bleeding time, tendency to bleedings).
The benzalkoniya chloride (preservative) which can be laid for surfaces of soft contact lenses is a part of drug therefore lenses need to be removed in 15 minutes prior to burying, their carrying is possible not earlier than 15 minutes after burying.
Use in pediatrics
Safety of use of eye drops Diclofenac at children is not established. Appointment is possible only in cases when the expected advantage of therapy exceeds risk of use of drug for this category of patients.
Pregnancy and a lactation
of Adequate and strictly controlled researches of safety of use at pregnancy and in the period of a lactation (breastfeeding) it is not carried out. Use of drug for this category of patients is possible only if the expected advantage of therapy for mother exceeds potential risk for a fruit or the baby.
Influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
does not influence.
Patients at whom after applique the sight clearness is temporarily lost are not recommended to drive or work with a difficult technique right after burying drug.
The overdose
At use of eye drops Diclofenac in the recommended doses
overdose is improbable. Now about cases of overdose of the drug Diclofenac it was not reported.
Overdose symptoms: strengthening of side effects of drug.
Treatment: to wash eyes a large number with warm water.
A form of release and packing
Eye drops, solution of 0.1% of diclofenac of sodium
On 5 ml in a white bottle from polyethylene with a stopper dropper and the screwing-up protective cap supplied with a safety ring.
1 bottle together with the instruction for use in a cardboard pack.
To Store storage conditions at a temperature from + 150C to + 250C in original packing.
To store out of children's reach!
A period of storage
3 years
do not use drug after the expiry date specified on packing.
After opening the bottle should be stored in the fridge within 28 days.
Prescription status
According to the prescription
of Proizvoditel Rompharm Company of S.R. L.,
Romania Eroilor St. 1A, Otopen.
ph. +40 21 208 9743, +40 21 208 97 42, fax: +40 21 266 49 38