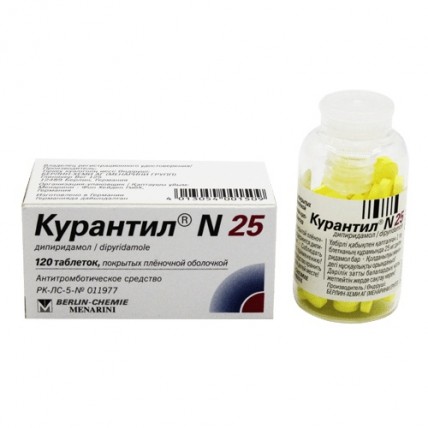
Curantil® (Dipyridamole) 25 mg, 120 tablets
- $19.10
What is Curantyl® N 25?
Curantyl® N 25 is a medicine with the active ingredient dipyridamole, used primarily to prevent blood clots. It comes in film-coated tablets with a dosage of 25 mg. Dipyridamole works by inhibiting platelet aggregation, which helps to reduce the risk of thromboembolism, particularly after heart valve replacement surgery when combined with oral anticoagulants.
How Should Curantyl® N 25 Be Taken?
What is the Recommended Dosage?
The optimal dose of Curantyl® N 25 for adults is between 300-600 mg per day, divided into 3 or 4 doses. It is important to follow the dosage instructions provided by your healthcare provider, as the exact dose may vary depending on your medical condition.
How Should the Tablets Be Taken?
Curantyl® N 25 tablets should be taken on an empty stomach, swallowed whole with plenty of water. Do not chew or break the tablets. The treatment duration and dosage will be determined by your healthcare provider based on your individual needs and how well you tolerate the medicine.
What Precautions Should Be Taken Before Using Curantyl® N 25?
Who Should Not Take This Medicine?
Do not take Curantyl® N 25 if you have:
- Hypersensitivity to dipyridamole or any other component of the medicine.
- Recently had a myocardial infarction.
- Unstable angina.
- Severe atherosclerosis of coronary arteries.
- Subaortic stenosis.
- Decompensated heart failure.
- Severe blood pressure problems or hypotension.
- Severe cardiac arrhythmias.
- Hemorrhagic diathesis.
- Conditions prone to bleeding, such as peptic ulcers.
- Are under 18 years of age.
- Are breastfeeding.
- Have hereditary intolerance to fructose or galactose.
What Are the Necessary Precautions?
- Dipyridamole acts as a vasodilator, so caution is required in patients with severe ischemic heart disease, including those with unstable angina or recent myocardial infarction, left ventricular outflow obstruction, or hemodynamic instability.
- Patients regularly taking Curantyl® N 25 should avoid additional intravenous dipyridamole.
- Caution is advised for patients with bleeding disorders or those on anticoagulants or antiplatelet medications.
Can Curantyl® N 25 Interact with Other Medicines?
Yes, Curantyl® N 25 can interact with other medications:
- It increases the plasma levels and cardiovascular effects of adenosine, so the dose of adenosine may need adjustment.
- Antacids may reduce the effectiveness of Curantyl® N 25.
- There is an additive effect with aspirin on platelet function.
- It may enhance the effects of oral anticoagulants.
- Dipyridamole may reduce the absorption of fludarabine and increase the absorption of digoxin.
- Xanthine derivatives, such as those found in coffee and tea, can weaken the vasodilatory effect of Curantyl® N 25.
Are There Any Side Effects Associated with Curantyl® N 25?
Common side effects include:
- Headache and dizziness.
- Nausea and diarrhea.
- Angina.
- Hypotension and flushing.
- Vomiting and rash.
- Muscle pain (myalgia).
Rare side effects may include:
- Hypersensitivity reactions.
- Thrombocytopenia.
- Tachycardia and exacerbation of ischemic heart disease.
- Bronchospasm and angioedema.
- Increased bleeding tendency during or after surgery.
How Should Curantyl® N 25 Be Stored?
Curantyl® N 25 should be stored at a temperature not exceeding 25°C (77°F) and kept out of reach of children. Do not use the medicine after the expiry date.
What Should Be Done in Case of an Overdose?
Symptoms of overdose may include:
- Vasodilation and hypotension.
- Angina-like symptoms.
- Tachycardia, feeling of warmth, facial flushing, weakness, dizziness, and anxiety.
Treatment for overdose includes:
- Symptomatic therapy for oral intoxication, such as gastric lavage or induced vomiting.
- Slow intravenous administration of aminophylline (50-100 mg over 60 seconds) to counteract the vasodilatory effects.
- Sublingual nitroglycerin if angina symptoms persist.
For any questions about the use of Curantyl® N 25, consult your healthcare provider.