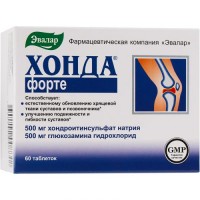
Chondroxide 250 mg (60 tablets)
- $50.80
The instruction for medical use
of Hondroksid® medicine
the Trade name
of Hondroksid®
the International unlicensed name
Is not present
the Dosage form
of the Tablet of 250 mg
Structure
One tablet contains
active agent - chondroitin sodium sulfate of 250 mg,
excipients: calcium stearate, krospovidon, K-30 povidone, cellulose microcrystalline, hydroxycarbonate magnesium pentahydrate.
The description
of the Tablet of round shape, with a flat surface, from color, white to white with a yellowish or creamy shade, with risky and a facet, is allowed existence of impregnations
Pharmacotherapeutic group
Anti-inflammatory and antirheumatic drugs. Non-steroidal anti-inflammatory drugs. Other non-steroidal anti-inflammatory drugs. Chondroitin sulfate
the ATX M01AX25 Code
the Pharmacological
Pharmacokinetics Absorption At properties single dose in a sredneterapevtichesky dose the maximum concentration in blood plasma is reached in 3-4 hours, in synovial fluid – in 4-5 hours. The bioavailability of drug is 13%.
Distribution
Collects, mainly, in cartilaginous tissue (the maximum concentration in an articulate cartilage is reached in 48 hours), the synovial membrane is not an obstacle for its penetration into a joint cavity.
Removal
Is removed by kidneys during 24 h.
The pharmacodynamics
of Hondroksid® has the hondrostimuliruyushchy, regenerating and anti-inflammatory effect, participates in creation of the main substance of a cartilaginous and bone tissue, influences exchange processes in hyaline and fibrous cartilages. Slows down processes of a degeneration (destruction) of cartilaginous tissue and stimulates recovery processes in an articulate cartilage.
Хондроксид® stimulates biosynthesis of proteoglycans, promotes regeneration of an articulate bag and the cartilaginous surfaces of joints, has anti-inflammatory effect. Slows down a resorption of a bone tissue and reduces calcium losses, accelerates processes of recovery of a bone tissue.
Хондроксид® slows down progressing of an osteoarthrosis and osteochondrosis. At use of the drug Hondroksid® the morbidity decreases and the mobility of the affected joints improves, at the same time the therapeutic effect remains a long time after the termination of a course of therapy.
Having structural similarity with heparin, can potentially interfere with formation of fibrinous blood clots in a synovial and subchondral microcirculation.
Indications
- degenerative dystrophic diseases of joints and a backbone (osteoarthrosis, including an osteoarthrosis of a temporal and mandibular joint, a spondylarthrosis, osteochondrosis, osteoporosis, heyroartropatiya)
the Route of administration and doses
Inside, on 0.5 g (2 tablets) 2 times a day.
A pill is taken, washing down with a small amount of water.
The recommended duration of an initial course is 6 months. It is necessary to consider that therapeutic effect of drug remains within 3-5 months after its cancellation depending on localization and a stage of a disease. If necessary carrying out repeated courses of treatment which duration is defined individually is possible.
Side effects
Seldom (from & gt, 0.01% to & lt, 0.1%)
- allergic reactions
- nausea, vomiting, a constipation, pains in epigastriums, a meteorism,
Contraindication diarrhea
- hypersensitivity to drug components
- children's and teenage age up to 18 years
- pregnancy
- the lactation period
- tendency to bleeding, thrombophlebitises
- the profound renal failures
Medicinal interactions
strengthening of effect of indirect anticoagulants, antiagregant, fibrinolitik is possible.
Special instructions
At combined use of drug with indirect anticoagulants, antiagregant, fibrinolitika more frequent control of indicators of fibrillation is required. It is not applied in bleedings or tendency to them.
The feature of influence of medicine on ability to drive the car or potentially dangerous mechanisms
does not affect ability to run the vehicle or potentially dangerous mechanisms.
Overdose
Symptoms: nausea, vomiting, diarrhea, at long reception in excessively high doses (more than 3 g/days) are possible hemorrhagic rashes.
Treatment: performing symptomatic therapy.
The form of release and packing
On 10 tablets place in blister strip packaging from a film polyvinylchloride either polyvinylchloride/polyvinylidene chloride or polyvinylchloride/polyethylene/polyvinylidene chloride or laminate the focused polyamide/aluminium/polyvinylchloride and the printing aluminum foil varnished.
On 6 planimetric packs together with the instruction for medical use in the state and Russian languages place in a pack from cardboard.
To Store storage conditions in the dry, protected from light place at a temperature not above 25 °C.
To store out of children's reach!
2 years
not to apply a period of storage after an expiration date.
Prescription status
Without prescription
JSC Nizhpharm Producer,
Russian Federation 603950, Nizhny Novgorod, GSP-459, Salganskaya St., 7
ph.: (831) 278-80-88
fax: (831) 430-72-28
a web the website: http://www.nizhpharm.ru
the Name and the country of the owner of the registration certificate
of JSC Nizhpharm, the Russian Federation
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products (goods)
Representative office of JSC Nizhpharm
050043, Republic of Kazakhstan, Almaty, mdt. Khan-Taniri, 55b
ph.: (727) 398-64-91, 398-64-92, 398-64-93, 243-45-03, 243-45-04
fax: (727) 398-64-95
e–mail: almaty@stada.kz
of Hondroksid® medicine
the Trade name
of Hondroksid®
the International unlicensed name
Is not present
the Dosage form
of the Tablet of 250 mg
Structure
One tablet contains
active agent - chondroitin sodium sulfate of 250 mg,
excipients: calcium stearate, krospovidon, K-30 povidone, cellulose microcrystalline, hydroxycarbonate magnesium pentahydrate.
The description
of the Tablet of round shape, with a flat surface, from color, white to white with a yellowish or creamy shade, with risky and a facet, is allowed existence of impregnations
Pharmacotherapeutic group
Anti-inflammatory and antirheumatic drugs. Non-steroidal anti-inflammatory drugs. Other non-steroidal anti-inflammatory drugs. Chondroitin sulfate
the ATX M01AX25 Code
the Pharmacological
Pharmacokinetics Absorption At properties single dose in a sredneterapevtichesky dose the maximum concentration in blood plasma is reached in 3-4 hours, in synovial fluid – in 4-5 hours. The bioavailability of drug is 13%.
Distribution
Collects, mainly, in cartilaginous tissue (the maximum concentration in an articulate cartilage is reached in 48 hours), the synovial membrane is not an obstacle for its penetration into a joint cavity.
Removal
Is removed by kidneys during 24 h.
The pharmacodynamics
of Hondroksid® has the hondrostimuliruyushchy, regenerating and anti-inflammatory effect, participates in creation of the main substance of a cartilaginous and bone tissue, influences exchange processes in hyaline and fibrous cartilages. Slows down processes of a degeneration (destruction) of cartilaginous tissue and stimulates recovery processes in an articulate cartilage.
Хондроксид® stimulates biosynthesis of proteoglycans, promotes regeneration of an articulate bag and the cartilaginous surfaces of joints, has anti-inflammatory effect. Slows down a resorption of a bone tissue and reduces calcium losses, accelerates processes of recovery of a bone tissue.
Хондроксид® slows down progressing of an osteoarthrosis and osteochondrosis. At use of the drug Hondroksid® the morbidity decreases and the mobility of the affected joints improves, at the same time the therapeutic effect remains a long time after the termination of a course of therapy.
Having structural similarity with heparin, can potentially interfere with formation of fibrinous blood clots in a synovial and subchondral microcirculation.
Indications
- degenerative dystrophic diseases of joints and a backbone (osteoarthrosis, including an osteoarthrosis of a temporal and mandibular joint, a spondylarthrosis, osteochondrosis, osteoporosis, heyroartropatiya)
the Route of administration and doses
Inside, on 0.5 g (2 tablets) 2 times a day.
A pill is taken, washing down with a small amount of water.
The recommended duration of an initial course is 6 months. It is necessary to consider that therapeutic effect of drug remains within 3-5 months after its cancellation depending on localization and a stage of a disease. If necessary carrying out repeated courses of treatment which duration is defined individually is possible.
Side effects
Seldom (from & gt, 0.01% to & lt, 0.1%)
- allergic reactions
- nausea, vomiting, a constipation, pains in epigastriums, a meteorism,
Contraindication diarrhea
- hypersensitivity to drug components
- children's and teenage age up to 18 years
- pregnancy
- the lactation period
- tendency to bleeding, thrombophlebitises
- the profound renal failures
Medicinal interactions
strengthening of effect of indirect anticoagulants, antiagregant, fibrinolitik is possible.
Special instructions
At combined use of drug with indirect anticoagulants, antiagregant, fibrinolitika more frequent control of indicators of fibrillation is required. It is not applied in bleedings or tendency to them.
The feature of influence of medicine on ability to drive the car or potentially dangerous mechanisms
does not affect ability to run the vehicle or potentially dangerous mechanisms.
Overdose
Symptoms: nausea, vomiting, diarrhea, at long reception in excessively high doses (more than 3 g/days) are possible hemorrhagic rashes.
Treatment: performing symptomatic therapy.
The form of release and packing
On 10 tablets place in blister strip packaging from a film polyvinylchloride either polyvinylchloride/polyvinylidene chloride or polyvinylchloride/polyethylene/polyvinylidene chloride or laminate the focused polyamide/aluminium/polyvinylchloride and the printing aluminum foil varnished.
On 6 planimetric packs together with the instruction for medical use in the state and Russian languages place in a pack from cardboard.
To Store storage conditions in the dry, protected from light place at a temperature not above 25 °C.
To store out of children's reach!
2 years
not to apply a period of storage after an expiration date.
Prescription status
Without prescription
JSC Nizhpharm Producer,
Russian Federation 603950, Nizhny Novgorod, GSP-459, Salganskaya St., 7
ph.: (831) 278-80-88
fax: (831) 430-72-28
a web the website: http://www.nizhpharm.ru
the Name and the country of the owner of the registration certificate
of JSC Nizhpharm, the Russian Federation
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products (goods)
Representative office of JSC Nizhpharm
050043, Republic of Kazakhstan, Almaty, mdt. Khan-Taniri, 55b
ph.: (727) 398-64-91, 398-64-92, 398-64-93, 243-45-03, 243-45-04
fax: (727) 398-64-95
e–mail: almaty@stada.kz