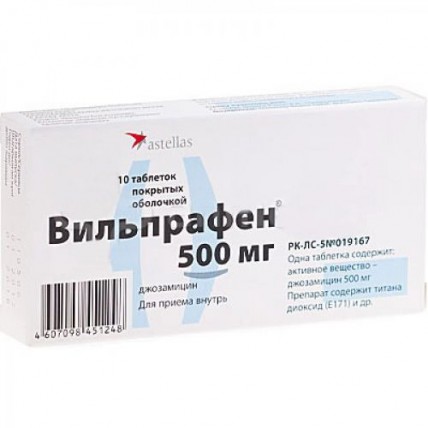
Vilprafen 10s 500 mg coated tablets
- $27.70
Out Of Stock
The instruction for medical use of Vilprafen Torgovoye medicine a name Vilprafen Mezhdunarodnoye the unlicensed name Dzhozamitsin Lekarstvennaya the Tablet form, coated, 500 mg Structure One tablet contains active agent - dzhozamitsin 500 mg, excipients: microcrystalline cellulose, polysorbate 80, silicon dioxide colloidal anhydrous, sodium carboxymethylcellulose, magnesium stearate structure of a cover: methyl cellulose, polyethyleneglycol 6000, talc, titan dioxide (E 171), aluminum hydroxide, copolymer of methacrylic acid and its ethers. The description of the Tablet, coated, white or almost white color, the oblong, biconvex, having risks on both sides. Pharmacotherapeutic group Antibacterial drugs for system use. Macroleads, linkozamida and streptogramina. Macroleads. Dzhozamitsin the ATX J01FA07 Code the Pharmacological Pharmacokinetics Later properties of intake dzhozamitsin is quickly and completely soaked up from digestive tract, meal does not affect bioavailability. The maximum concentration of a dzhozamitsin in serum is reached in 1-2 h after reception. About 15% of a dzhozamitsin contact proteins of plasma. High concentrations of substance are found in lungs, tonsils, saliva, sweat and the lacrimal liquid. Concentration in a phlegm exceeds concentration in plasma by 8-9 times. Collects in a bone tissue. There passes the placental barrier, cosecretes in breast milk. Dzhozamitsin is metabolized in a liver to less active metabolites and excreted mainly with bile. Excretion of drug with urine less than 20%. The pharmacodynamics Drug is used for treatment of bacterial infections, the bacteriostatic activity of a dzhozamitsin, as well as other macroleads, is caused by inhibition of synthesis of protein of bacteria. During creation in the center of inflammation of high concentrations has bactericidal effect. Dzhozamitsin is active concerning intracellular microorganisms (Chlamydia trachomatis and Chlamydia pneumoniae, Mycoplasma pneumoniae, Mycoplasma hominis, Ureaplasma urealyticum, Legionella pneumophila), gram-positive bacteria (Staphylococcus aureus, Streptococcus pyogenes and Streptococcus pneumoniae (pneumococcus), Corynebacterium diphteriae), gram-negative bacteria (Neisseria meningitidis, Neisseria gonorrhoeae, Haemophilus influenzae, Bordetella pertussis) and also against some anaerobic bacteria (Peptococcus, Peptostreptococcus, Clostridium perfringens). Slightly influences enterobakteriya therefore changes natural bacterial flora of digestive tract a little. It is effective at resistance to erythromycin. The resistance to a dzhozamitsin develops less than to other antibiotics from group of macroleads. Indications the Acute and persistent infections caused by microorganisms, sensitive to drug: - dental infections - infections of ENT organs - respiratory infections - mastitis - infections of soft tissues - infections of genitals - infections of bilious ways - at allergic reactions to penicillin the Route of administration and doses of the Tablet Vilprafen® should be swallowed entirely, washing down with a small amount of water. Adults and children are more senior than 14 years - from 1 to 2 g of a dzhozamitsin. The daily dose has to be divided into 2-3 receptions. The initial recommended dose makes from 1 g a day. Increase in a dose up to 3 g a day is in case of need allowed. To children aged from 6 years up to 14 years – appoint 30 - 50 mg/kg of mass of bodies/days in a dose, having divided into 3 receptions. In case of ordinary and spherical eels it is recommended to appoint Vilprafen® in a dose of 500 mg twice a day within the first 2-4 weeks, further - 500 mg as the supporting treatment within 8 weeks once a day. Usually duration of treatment is defined by the doctor. The course of treatment needs to be continued within 48 hours after disappearance of symptoms and normalization of temperature. The course of treatment of a staphylococcal infection should not exceed 10 days without the subsequent consultation of the doctor. Side effects As well as in a case with other macroleads, were observed allergic reactions of easy degree (small tortoiseshell and other types of rash) and a digestive disturbance of easy degree (anorexia, nausea, vomiting, a stomach ache, diarrhea). Control of function of a liver found several cases of gepatobiliarny insufficiency of easy degree which were reversible at the treatment termination. It was reported about several cases of a face edema. Frequency of the undesirable phenomena is distributed in the following look: very frequent (≥1/10), frequent (≥1/100, & lt, 1/10), infrequent (≥1/1000, & lt, 1/100), rare (≥1/10,000, & lt, 1/1000), very rare (& lt, 1/10,000), unknown (it is impossible to estimate on the available data). The following undesirable phenomena were registered during clinical trials and/or post-marketing experience. It is unknown: - an abdominal distension, stomatitis, nausea, a stomach ache and diarrhea, loss of appetite, pseudomembranous colitis - a purpura, bullous dermatitis, a multiformny erythema, Stephens-Johnson's syndrome, a small tortoiseshell and skin reactions, a Quincke's disease, a toxic epidermal necrolysis - anaphylactic reaction - an abnormal liver function and jaundice which usually includes (but it is not limited) the cholestatic reactions specifying an abnormal liver function of average degree. Contraindications - hypersensitivity to active agent and other components of drug or to chemically related substances - a serious gepatobiliarny illness Medicinal interactions Dzhozamitsin can influence metabolism of the antihistaminic drugs containing terfenadin or astemizol, vyzvy relative overdose considerably. Therefore simultaneous use of a terfenadin and dzhozamitsin it is necessary to avoid (see the section Special Instructions). There is a proof about increase in level of theophylline in blood plasma after reception of a dzhozamitsin at children. Increase in level of theophylline can be connected with increase in time of semi-removal. At detection the toxic level of theophylline it is necessary to modify a dose of theophylline and further to control theophylline level in plasma. Simultaneous use with alkaloids of an ergot can lead to vasoconstriction. If any symptoms or symptoms of poisoning an ergot were found it is necessary to stop treatment dzhozamitsiny and to begin the corresponding therapy (topical administration of heat and vasodilating drugs). Simultaneous use of a dzhozamitsin and cyclosporine / takrolimusa can cause increase in level of cyclosporine / takrolimusa to nephrotoxic level. It is regularly necessary to control cyclosporine level / takrolimusa in plasma. Dzhozamitsin can increase drowsiness after use of a triazolam. In case of overdose of a triazolam it is necessary to suspend its use. Dzhozamitsin can strengthen effect of Bromocriptinum mezilat and cause drowsiness, dizziness, an ataxy, etc. In case of poisoning with Bromocriptinum mezilat it is necessary to suspend its use. Dzhozamitsin can strengthen effect of oral anticoagulants, such as warfarin and its derivatives of vitamin K. Dzhozamitsin reduces production of vitamin K colibacilli. In case of bleeding, treatment with dzhozamitsiny and/or oral anticoagulants and vitamin K has to be suspended, depending on weight of the bleeding or a class of a coagulopathy based on PTV/MNO. Special instructions At patients with a liver failure the increased drug level in plasma can be noted. Thus, it is necessary to be careful at use of a dzhozamitsin for patients with a liver failure. In general diseases when treatment lasts longer, than 15 days are recommended to control function of a liver at patients with gepatobiliarny. Though there are no convincing proofs, it is impossible to exclude that treatment (especially long-term treatment) dzhozamitsiny and other makrolidny antibiotics can lead to distribution of resistant bacteria and mushrooms. In such cases the treatment has to be it is stopped and is necessary to begin the corresponding treatment. It is necessary to show care at simultaneous use of a dzhozamitsin with the antihistaminic drugs containing terfenadin or astemizol as dzhozamitsin reduces removal of these drugs that can lead to serious violations of a warm rhythm. Pregnancy and a lactation Exists limited data on use of a dzhozamitsin for pregnant women. Thus, at women who are or can be pregnant women, drug should be used only under careful medical observation and only when its therapeutic advantage exceeds possible risks. As dzhozamitsin it is allocated with breast milk, the feeding women have to consult with the doctor. The feature of influence of drug on ability to run motor transport and other potentially dangerous mechanisms does not affect ability to drive the car and to operate difficult mechanisms. Overdose Symptoms: strengthening of side effects, especially from digestive tract, such as nausea or diarrhea. Treatment: symptomatic. A form of release and packing On 10 tablets in blister strip packaging from a film of polyvinylchloride and aluminum foil. On 1 planimetric packing together with the instruction for medical use in the state and Russian languages place in a pack cardboard. Storage conditions In the place protected from light, at a temperature not over 25 ºС. To store out of children's reach. 4 years not to apply a period of storage after the period of storage specified on packing. Prescription status According to the prescription the Name and the country of manufacturing organization Dragenofarm Apotheker Pyushl GmbH, Gelstrasse 1, 84529, Titmoning, Germany the Name and the country of the owner of the registration certificate Astellas of Pharm B.V. Europe, the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of the products (goods) responsible for post-registration observation of safety of medicine Representation Astellas of Pharm B.V. Europe in PK 050059, Republic of Kazakhstan, Almaty, Al-Farabi Ave. 15, BC Nurly Tau, body 4B, office No. 20
to Develop Netherlands Silviusveg 62, 2333 VE Leiden, Netherlands
to Develop Netherlands Silviusveg 62, 2333 VE Leiden, Netherlands