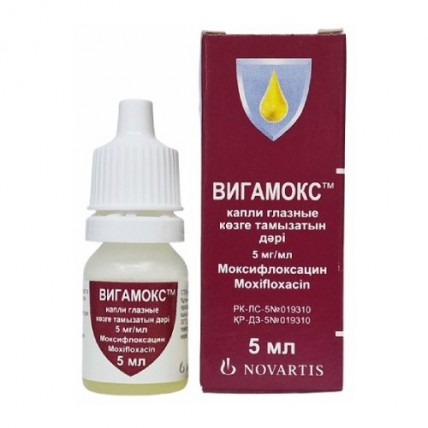
VIGAMOX® (Moxifloxacin) Eye Drops 0.5%, 5 ml
- $27.00
What is Vigamox® (Moxifloxacin) Eye Drops 0.5%?
Vigamox® (moxifloxacin hydrochloride ophthalmic solution) 0.5% is a sterile, topical solution used to treat bacterial eye infections. It contains moxifloxacin, a fourth-generation fluoroquinolone antibiotic that is highly effective against a wide range of bacterial strains, including both Gram-positive and Gram-negative bacteria, as well as atypical microorganisms and anaerobes. Vigamox® is widely used for treating conditions such as bacterial conjunctivitis and other ocular infections caused by susceptible bacteria. The solution is self-preserved, meaning it does not contain preservatives, which makes it suitable for individuals with sensitive eyes.
How does Vigamox® work?
Vigamox® contains moxifloxacin, which works by inhibiting bacterial enzymes that are crucial for the replication and repair of bacterial DNA. Specifically, it targets topoisomerase II (DNA gyrase) and topoisomerase IV, which bacteria need for their survival. By inhibiting these enzymes, moxifloxacin effectively halts bacterial growth and kills the infection-causing microorganisms.
One of the unique features of moxifloxacin is its advanced chemical structure. It has a C8-methoxy group, which reduces the selection of resistant bacterial mutants, and a bulky C-7 substituent group that helps prevent bacterial efflux pumps from ejecting the antibiotic. These properties make Vigamox® more effective in treating infections caused by resistant bacteria.
What bacteria does Vigamox® target?
Vigamox® is effective against a wide range of bacterial pathogens, including many that are resistant to other antibiotics.
Below is a detailed breakdown of the bacteria that Vigamox® targets:
Aerobic Gram-positive Bacteria:
- Corynebacterium species: Common in ocular infections, especially in immunocompromised patients.
- Micrococcus luteus: Effective against strains resistant to antibiotics like erythromycin, gentamicin, tetracycline, and trimethoprim.
- Staphylococcus aureus: Including methicillin-resistant Staphylococcus aureus (MRSA), which is often resistant to several antibiotics.
- Staphylococcus epidermidis: Effective against methicillin-resistant strains that cause eye infections.
- Staphylococcus haemolyticus: Often resistant to methicillin and other antibiotics.
- Staphylococcus hominis: A frequent cause of ocular infections, especially in vulnerable populations.
- Staphylococcus warneri: Known for its resistance to erythromycin.
- Streptococcus pneumoniae: Including penicillin-resistant strains, a common cause of conjunctivitis.
- Streptococcus viridans group: Includes species resistant to penicillin and other antibiotics.
Aerobic Gram-negative Bacteria:
- Acinetobacter lwofii: Associated with hospital-acquired infections, especially in immunocompromised individuals.
- Haemophilus influenzae: Including ampicillin-resistant strains, commonly causing conjunctivitis.
- Haemophilus parainfluenzae: Responsible for various ocular infections.
Other Pathogens:
- Chlamydia trachomatis: Causes trachoma and adult inclusion conjunctivitis.
What other bacteria show in vitro activity to Vigamox®?
In laboratory studies, moxifloxacin has been shown to be active against additional bacterial strains.
While these are not typically the primary targets of Vigamox®, the drug has demonstrated the ability to inhibit these bacteria in vitro:
Aerobic Gram-positive Bacteria:
- Listeria monocytogenes: Can cause serious infections in immunocompromised individuals.
- Staphylococcus saprophyticus: A common pathogen in urinary tract infections, but also capable of causing eye infections.
- Streptococcus agalactiae: Known for neonatal infections but can also infect the eyes.
- Streptococcus mitis: A member of the viridans group that can cause ocular infections.
- Streptococcus pyogenes: Causes serious infections like conjunctivitis.
Aerobic Gram-negative Bacteria:
- Acinetobacter baumannii: A multi-drug resistant bacterium notorious for hospital-acquired infections.
- Citrobacter freundii, Citrobacter koseri, Enterobacter aerogenes, and Enterobacter cloacae: These bacteria are often found in hospital settings and can cause serious infections.
- Escherichia coli: Certain strains can cause eye infections.
- Klebsiella pneumoniae: Often resistant to antibiotics, causing ocular infections.
- Neisseria gonorrhoeae: Responsible for severe conjunctivitis, especially in newborns.
Anaerobic Bacteria:
- Clostridium perfringens: Can cause serious infections, including rare cases of ocular involvement.
- Prevotella species: Known for their involvement in periodontal disease but also capable of causing eye infections.
Other Microorganisms:
- Chlamydia pneumoniae: Causes respiratory infections but can also affect the eyes.
- Legionella pneumophila: While typically a respiratory pathogen, it can also infect the eyes.
- Mycoplasma pneumoniae: Known for respiratory infections but also capable of causing conjunctivitis.
What are the benefits of Vigamox® eye drops?
- Broad-spectrum antibacterial action: Vigamox® is effective against a wide range of bacterial pathogens, including many resistant strains.
- Self-preserved formula: The solution does not contain preservatives, which makes it less likely to cause irritation in people with sensitive eyes.
- Convenient dosing regimen: Only three applications per day are needed, making it easy to comply with the treatment.
- Rapid relief: Vigamox® quickly alleviates the symptoms of bacterial eye infections such as redness, discharge, and discomfort.
How to use Vigamox® (Moxifloxacin) Eye Drops?
Vigamox® is for topical use only. The standard dosage is one drop into the affected eye(s) three times a day for a course of four days. Follow your doctor’s recommendations carefully and complete the full course of treatment, even if your symptoms improve earlier.
- If you are using multiple eye medications, make sure to wait at least five minutes between applying different medications.
- Avoid contact lenses: It is recommended to avoid wearing contact lenses during treatment, especially in cases of active infection.
Who can use Vigamox®?
Vigamox® is safe for most adults and children. It does not require any dosage adjustment for elderly patients or for those with kidney or liver impairments, as systemic absorption through the eye is minimal. However, individuals with a known allergy to moxifloxacin or other quinolone antibiotics should avoid using Vigamox®.
What precautions should you take when using Vigamox®?
- Allergic reactions: If you notice any signs of hypersensitivity, such as swelling, itching, or severe redness, discontinue use and consult a healthcare professional immediately.
- Do not inject: Vigamox® is intended for topical use only and should never be injected into the eye.
- Avoid contamination: Do not touch the dropper tip to any surface, including your eye, to avoid contamination of the solution.
What are the possible side effects of Vigamox®?
Most people tolerate Vigamox® well, but some may experience mild side effects, including:
- Temporary eye irritation or discomfort
- Dryness or a burning sensation in the eyes
- Blurred vision (temporarily)
Less common side effects may include eye redness, itching, or swelling of the eyelids. Severe allergic reactions are rare but require immediate medical attention.
How to store Vigamox®?
To maintain its effectiveness, store Vigamox® at room temperature, no higher than 30°C (86°F).
After opening, the product should be used within four weeks.
Make sure to keep the bottle tightly closed and out of reach of children.