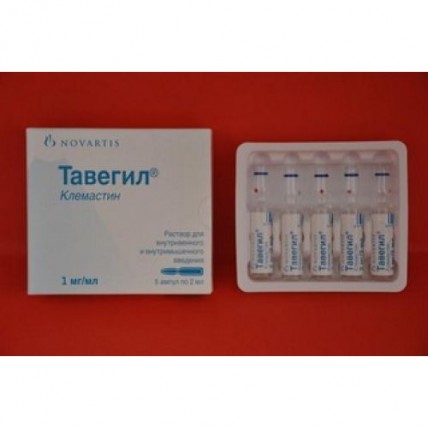
TAVEGYL® (Clemastine) 1 mg/ml, 2 ml x 5 amps solution for injection
- $11.00
Out Of Stock
The instruction for use of medicine for experts
TAVEGILT (TAVEGYLT)
the Trade name
of TAVEGILT
the International unlicensed
name Clemastinum Dosage Form Solution for intravenous and intramuscular administration, 2 ml
Structure
of 2 ml of solution contain
active agent - 2.68 mg of Clemastinum of the hydrofumarat (2 mg of Clemastinum),
excipients: sorbitol, ethanol, propylene glycol, sodium citrate, water for injections.
Description
Transparent, colourless or very pale yellow or very pale chartreuse solution
Pharmacotherapeutic group
Antihistamines of systemic action.
Aminoalkyl ethers
the Code of automatic telephone exchange R06AA04
the Pharmacological
Pharmacokinetics Communication properties with proteins of plasma makes 95%.
In the period of a lactation a small amount of Clemastinum can get into breast milk.
Clemastinum is exposed to considerable metabolism in a liver. Metabolites generally (45-65%) are removed through kidneys with urine, not changed active agent is found in urine only in trace quantities.
The pharmacodynamics
of Tavegil® is an antagonist of H1 receptors.
Treats antihistamines from group of benzgidrilny ethers, reduces permeability of capillaries, has the antiallergenic and antipruritic effect which is characterized by quick start and considerable duration of action.
Indications
- prevention or treatment of allergic and pseudo-allergic reactions (including at administration of contrast agents, blood transfusion, diagnostic use of a histamine)
- a Quincke's disease
- acute or anaphylactoid anaphylaxis (as an additional tool).
Route of administration and doses
Adult: the average dose makes 2 mg (2 ml), that is contents 1 ampoules. The drug is administered intravenously or intramusculary.
With the preventive purpose the drug is administered intravenously struyno slowly in a dose of 2 mg just before possible emergence of anaphylactic reaction or reaction in response to use of a histamine. Solution of drug can be in addition divorced with isotonic solution of sodium of chloride or 5% dextrose solution in the ratio to 1:5
Children: 0.025 in mg/kg a day intramusculary for 2 injections.
Side effects
- increased fatigue, drowsiness, a headache, dizziness, a tremor, sedation, weakness, feeling of fatigue, block, a lack of coordination of movements
- it is rare (especially at children): the concern, acrimony, excitement, nervousness, insomnia, hysteria, euphoria, spasms, a parasthesia
- dyspepsia, nausea, pains in epigastriums, a constipation, vomiting
- seldom dryness in a mouth, a loss of appetite and diarrhea
- very seldom speeded up or complicated urination
- is rare: condensation of a bronchial secret and difficulty of an otkhozhdeniye of a phlegm, feeling of pressure in a thorax and breath difficulty, congestion of a nose
- is rare: the lowering of arterial pressure (is more often at elderly patients), heartbeat, premature ventricular contraction
- is very rare: urticaria, skin rash, photosensitization
acute anaphylaxis.
Contraindications
- hypersensitivity to Tavegilu®
- simultaneous use of inhibitors of a monoaminooxidase
- bronchial asthma
- age are younger than 1 year
- women during feeding by a breast.
Medicinal interactions
of Tavegil® effect of the medicines oppressing central nervous system (somnolent, sedative, anxiolytics) strengthens, M-holinoblokatorov and also alcohol. It is not compatible to a concomitant use of inhibitors of a monoaminooxidase.
Special instructions
Intra arterial administration of drug is not allowed!
It is necessary to observe extra care at use of Tavegila® for patients with the stenosing stomach ulcer, piloroduodenalny obstruction, with obstruction of a neck of a bladder and also the prostatauxe which is followed by an ischuria with the increased intraocular pressure, a hyperthyroidism, diseases of a cardiovascular system, including arterial hypertension.
Pregnancy and lactation
during pregnancy of Tavegil® apply only if the expected advantage for mother exceeds potential risk for a fruit.
The feature of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
the Patient accepting Tavegil® is recommended to abstain from driving of vehicles, work with mechanisms and also from
other types of activity demanding the increased concentration of attention and speed of psychomotor reactions.
Overdose
Symptoms. The overdose of antihistamines can lead both to oppressing, and to promoting effect on central nervous system. Stimulation of central nervous system is more often observed at children.
Also the phenomena of anticholinergic action can develop:
dryness in a mouth, the fixed expansion of pupils, rushes of blood to an upper half of a body, disturbance from digestive tract (nausea, pains in epigastriums, vomiting).
Treatment: Symptomatic therapy is shown.
A form of release and packing
of the Ampoule on 2 ml (1 mg/ml) from glass type 1.
On 5 ampoules together with the instruction for use pack into a cardboard box.
To Store storage conditions at a temperature of 15 - 30 0C
to Store out of children's reach!
Not to use an expiration date of 5 years after the term specified on packing.
Prescription status
According to the prescription
of Proizvoditel Novartis Konsyyumer Hels SA, Switzerland, it is made Nikomed Austria GmbH, Austria.
Address: St Peter Strasse 25, A-4020 Linz, Austria.
Novartis Consumer Health SA, Switzerland, produced Nycomed Austria GmbH, Austria.
Adress: St. - Peter-Strasse 25, A-4020 Linz, Austria.
Representation in
Russia 123104 Moscow, B. Palashevsky Lane, 15.
Ph. (095) 969-21-65, fax (095) 969-21-66
e-mail: consumerhealth@novartis.ru
TAVEGILT (TAVEGYLT)
the Trade name
of TAVEGILT
the International unlicensed
name Clemastinum Dosage Form Solution for intravenous and intramuscular administration, 2 ml
Structure
of 2 ml of solution contain
active agent - 2.68 mg of Clemastinum of the hydrofumarat (2 mg of Clemastinum),
excipients: sorbitol, ethanol, propylene glycol, sodium citrate, water for injections.
Description
Transparent, colourless or very pale yellow or very pale chartreuse solution
Pharmacotherapeutic group
Antihistamines of systemic action.
Aminoalkyl ethers
the Code of automatic telephone exchange R06AA04
the Pharmacological
Pharmacokinetics Communication properties with proteins of plasma makes 95%.
In the period of a lactation a small amount of Clemastinum can get into breast milk.
Clemastinum is exposed to considerable metabolism in a liver. Metabolites generally (45-65%) are removed through kidneys with urine, not changed active agent is found in urine only in trace quantities.
The pharmacodynamics
of Tavegil® is an antagonist of H1 receptors.
Treats antihistamines from group of benzgidrilny ethers, reduces permeability of capillaries, has the antiallergenic and antipruritic effect which is characterized by quick start and considerable duration of action.
Indications
- prevention or treatment of allergic and pseudo-allergic reactions (including at administration of contrast agents, blood transfusion, diagnostic use of a histamine)
- a Quincke's disease
- acute or anaphylactoid anaphylaxis (as an additional tool).
Route of administration and doses
Adult: the average dose makes 2 mg (2 ml), that is contents 1 ampoules. The drug is administered intravenously or intramusculary.
With the preventive purpose the drug is administered intravenously struyno slowly in a dose of 2 mg just before possible emergence of anaphylactic reaction or reaction in response to use of a histamine. Solution of drug can be in addition divorced with isotonic solution of sodium of chloride or 5% dextrose solution in the ratio to 1:5
Children: 0.025 in mg/kg a day intramusculary for 2 injections.
Side effects
- increased fatigue, drowsiness, a headache, dizziness, a tremor, sedation, weakness, feeling of fatigue, block, a lack of coordination of movements
- it is rare (especially at children): the concern, acrimony, excitement, nervousness, insomnia, hysteria, euphoria, spasms, a parasthesia
- dyspepsia, nausea, pains in epigastriums, a constipation, vomiting
- seldom dryness in a mouth, a loss of appetite and diarrhea
- very seldom speeded up or complicated urination
- is rare: condensation of a bronchial secret and difficulty of an otkhozhdeniye of a phlegm, feeling of pressure in a thorax and breath difficulty, congestion of a nose
- is rare: the lowering of arterial pressure (is more often at elderly patients), heartbeat, premature ventricular contraction
- is very rare: urticaria, skin rash, photosensitization
acute anaphylaxis.
Contraindications
- hypersensitivity to Tavegilu®
- simultaneous use of inhibitors of a monoaminooxidase
- bronchial asthma
- age are younger than 1 year
- women during feeding by a breast.
Medicinal interactions
of Tavegil® effect of the medicines oppressing central nervous system (somnolent, sedative, anxiolytics) strengthens, M-holinoblokatorov and also alcohol. It is not compatible to a concomitant use of inhibitors of a monoaminooxidase.
Special instructions
Intra arterial administration of drug is not allowed!
It is necessary to observe extra care at use of Tavegila® for patients with the stenosing stomach ulcer, piloroduodenalny obstruction, with obstruction of a neck of a bladder and also the prostatauxe which is followed by an ischuria with the increased intraocular pressure, a hyperthyroidism, diseases of a cardiovascular system, including arterial hypertension.
Pregnancy and lactation
during pregnancy of Tavegil® apply only if the expected advantage for mother exceeds potential risk for a fruit.
The feature of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
the Patient accepting Tavegil® is recommended to abstain from driving of vehicles, work with mechanisms and also from
other types of activity demanding the increased concentration of attention and speed of psychomotor reactions.
Overdose
Symptoms. The overdose of antihistamines can lead both to oppressing, and to promoting effect on central nervous system. Stimulation of central nervous system is more often observed at children.
Also the phenomena of anticholinergic action can develop:
dryness in a mouth, the fixed expansion of pupils, rushes of blood to an upper half of a body, disturbance from digestive tract (nausea, pains in epigastriums, vomiting).
Treatment: Symptomatic therapy is shown.
A form of release and packing
of the Ampoule on 2 ml (1 mg/ml) from glass type 1.
On 5 ampoules together with the instruction for use pack into a cardboard box.
To Store storage conditions at a temperature of 15 - 30 0C
to Store out of children's reach!
Not to use an expiration date of 5 years after the term specified on packing.
Prescription status
According to the prescription
of Proizvoditel Novartis Konsyyumer Hels SA, Switzerland, it is made Nikomed Austria GmbH, Austria.
Address: St Peter Strasse 25, A-4020 Linz, Austria.
Novartis Consumer Health SA, Switzerland, produced Nycomed Austria GmbH, Austria.
Adress: St. - Peter-Strasse 25, A-4020 Linz, Austria.
Representation in
Russia 123104 Moscow, B. Palashevsky Lane, 15.
Ph. (095) 969-21-65, fax (095) 969-21-66
e-mail: consumerhealth@novartis.ru