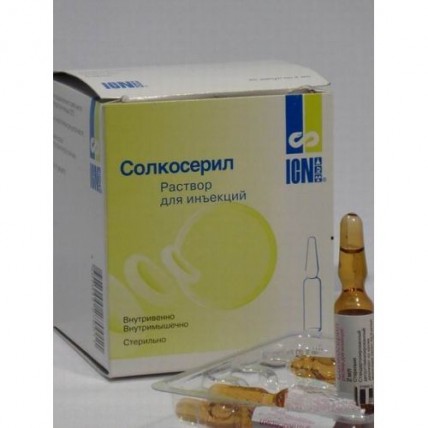
Solcoseryl 25's 2ml injection ampoules
- $166.50
Out Of Stock
Sku:
7ed56c936728
The instruction for medical use
of SOLKOSERIL medicine
the Trade name
of the drug Solkoseril Mezhdunarodnoye the unlicensed name
Is not present
the Dosage form
Solution for injections of 42.5 mg/ml
Structure
of 1 ml of solution contains
active agent - deproteined hemoderivative from blood of milk calfs (Bos taurus) standardized chemically and biologically (in terms of dry matter) 42.5 mg,
excipient water for injections to 1.0 ml, acid of chlorohydrogen 25% and/or sodium hydroxide of 27% (for finishing rn solution up to 7.0).
The description
Transparent solution from slightly yellowish till yellow color, with a slight smell of a solkoseril (the pleasant, reminding smell of beef-infusion broth).
The pharmacotherapeutic group
the Other hematologic
drugs ATX B06AB Code Pharmacological Pharmacokinetics Absorption Properties, distribution and elimination of active component of the drug Solkoseril of the deproteined standardized hemoderivative from blood of calfs (as well as pharmacokinetics of other biologically standardized drugs) does not give in to study by usual pharmacokinetic methods as it contains low-molecular components of plasma and uniform elements of blood which normal are present at an organism of people and animals.
In the course of studying pharmacokinetics it was revealed that drug begins to work, on average, in 20 min. (1030 min.) after an injection, and its action remains for 3 h after introduction.
The pharmacodynamics
Solkoseril is the deproteined hemoderivative from blood of milk calfs received by way of dialysis and ultrafiltration which contains a wide range of natural low-molecular substances with a molecular weight up to 5000 dalton and only some of them are described chemically and pharmacological. Solkoseril accelerates angenesis: normalizes and supports power metabolism and oxidizing phosphorylation, provides with cell phosphates, being in conditions of deficit of food, increases utilization of oxygen and stimulates glucose transport in the cells which are in conditions of a hypoxia and exhaustion of metabolic resources accelerates reparative and regenerative processes in the damaged fabrics, increases collagen synthesis, stimulates proliferation and migration of cells.
Thus, Solkoseril has protective action on cells in a condition of a hypoxia and metabolic disturbances, promotes angenesis, accelerates healing of wounds.
Indications
- occlusal diseases of peripheral arteries II IV stages according to Fontaine at patients with contraindications or intolerance of other medicines
- chronic venous insufficiency with
ulcers of shins, resistant to therapeutic influence,
the Route of administration and doses
Depending on disease severity the initial dose makes 10-20 ml intravenously in day, further - 5-10 ml a day intravenously or 2-5 ml intramusculary. At introduction in the form of infusions as solvent use 250 ml of isotonic solution of sodium chloride or 5% glucose solution.
At treatment of the occlusal diseases peripheral arteries (ODPA) - on 20 ml daily intravenously. Perhaps intravenous drop administration. Duration of therapy is up to 4 weeks and is defined by a clinical picture of a disease.
At treatment of the chronic venous insufficiency which is followed by trophic disturbances (Ulcera Cruris) on 10 ml intravenously 3 times a week. Duration of therapy is no more than 4 weeks and is defined by a clinical picture of a disease. The important additional action directed to prevention of peripheral hypostases (venous hypostases) is overlaying of a bandage using elastic roller.
By-effects
- hyperaemia and hypostasis in the place of an injection
- fervescence
Is rare
- allergic reactions (urticaria, an itching)
of the Contraindication
- hypersensitivity to drug components
- drug should not be used in the presence of allergic reaction
- children's age up to 18 years
Medicinal interactions
Solkoseril injections should not be mixed at introduction with other medicines, especially, with phytoextracts.
With care it is applied with the drugs increasing potassium level in blood (potassium drugs, kaliysberegayushchy diuretics, inhibitors of angiotensin-converting enzyme).
Special instructions
At intravenous use of not divorced drug it need to be entered slowly.
With care: hyperpotassemia, renal failure, disturbances of a warm rhythm, oligouriya, anury, fluid lungs, profound heart failure.
Pregnancy and the period of a lactation
Use of drug for treatment of pregnant women and the feeding women perhaps on doctor's orders if the expected advantage exceeds risk for a fruit and the child.
Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
the Overdose
Symptoms - strengthening of side effects does not influence.
Symptomatic treatment.
The form of release and packing
On 2 ml or 5 ml of drug place in ampoules of steady orange glass of I type neutral hydrolytic.
On 5 ampoules put in blister strip packaging.
On 5 planimetric packs together with the instruction for medical use in the state and Russian languages place in a pack from cardboard (for 2 ml).
On 1 planimetric packing together with the instruction for medical use in the state and Russian languages place in a pack from cardboard (for 5 ml).
To Store storage conditions at
a temperature not higher than 25 S. Hranit out of children's reach!
Not to apply a period of storage of 5 years after an expiration date.
Prescription status
According to the prescription
the Producer
of Legasi Pharmasyyutikals Switzerland Ryurbergstrasse
21, 4127 GmbH Birsfelden, Switzerland
the Owner of the registration certificate
of Pharmasyyutikals HONEY Switzerland GmbH, Switzerland
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products
Representation of Pharmasyyutikalz HONEY Switzerland GmbH in the Republic of Kazakhstan: Almaty, Dostyk Ave. 97, office 8, ph. +7 727 264-17-94, fax: + 7 727 264-17-71
E-mail address: info.safety@meda-cis.com
of SOLKOSERIL medicine
the Trade name
of the drug Solkoseril Mezhdunarodnoye the unlicensed name
Is not present
the Dosage form
Solution for injections of 42.5 mg/ml
Structure
of 1 ml of solution contains
active agent - deproteined hemoderivative from blood of milk calfs (Bos taurus) standardized chemically and biologically (in terms of dry matter) 42.5 mg,
excipient water for injections to 1.0 ml, acid of chlorohydrogen 25% and/or sodium hydroxide of 27% (for finishing rn solution up to 7.0).
The description
Transparent solution from slightly yellowish till yellow color, with a slight smell of a solkoseril (the pleasant, reminding smell of beef-infusion broth).
The pharmacotherapeutic group
the Other hematologic
drugs ATX B06AB Code Pharmacological Pharmacokinetics Absorption Properties, distribution and elimination of active component of the drug Solkoseril of the deproteined standardized hemoderivative from blood of calfs (as well as pharmacokinetics of other biologically standardized drugs) does not give in to study by usual pharmacokinetic methods as it contains low-molecular components of plasma and uniform elements of blood which normal are present at an organism of people and animals.
In the course of studying pharmacokinetics it was revealed that drug begins to work, on average, in 20 min. (1030 min.) after an injection, and its action remains for 3 h after introduction.
The pharmacodynamics
Solkoseril is the deproteined hemoderivative from blood of milk calfs received by way of dialysis and ultrafiltration which contains a wide range of natural low-molecular substances with a molecular weight up to 5000 dalton and only some of them are described chemically and pharmacological. Solkoseril accelerates angenesis: normalizes and supports power metabolism and oxidizing phosphorylation, provides with cell phosphates, being in conditions of deficit of food, increases utilization of oxygen and stimulates glucose transport in the cells which are in conditions of a hypoxia and exhaustion of metabolic resources accelerates reparative and regenerative processes in the damaged fabrics, increases collagen synthesis, stimulates proliferation and migration of cells.
Thus, Solkoseril has protective action on cells in a condition of a hypoxia and metabolic disturbances, promotes angenesis, accelerates healing of wounds.
Indications
- occlusal diseases of peripheral arteries II IV stages according to Fontaine at patients with contraindications or intolerance of other medicines
- chronic venous insufficiency with
ulcers of shins, resistant to therapeutic influence,
the Route of administration and doses
Depending on disease severity the initial dose makes 10-20 ml intravenously in day, further - 5-10 ml a day intravenously or 2-5 ml intramusculary. At introduction in the form of infusions as solvent use 250 ml of isotonic solution of sodium chloride or 5% glucose solution.
At treatment of the occlusal diseases peripheral arteries (ODPA) - on 20 ml daily intravenously. Perhaps intravenous drop administration. Duration of therapy is up to 4 weeks and is defined by a clinical picture of a disease.
At treatment of the chronic venous insufficiency which is followed by trophic disturbances (Ulcera Cruris) on 10 ml intravenously 3 times a week. Duration of therapy is no more than 4 weeks and is defined by a clinical picture of a disease. The important additional action directed to prevention of peripheral hypostases (venous hypostases) is overlaying of a bandage using elastic roller.
By-effects
- hyperaemia and hypostasis in the place of an injection
- fervescence
Is rare
- allergic reactions (urticaria, an itching)
of the Contraindication
- hypersensitivity to drug components
- drug should not be used in the presence of allergic reaction
- children's age up to 18 years
Medicinal interactions
Solkoseril injections should not be mixed at introduction with other medicines, especially, with phytoextracts.
With care it is applied with the drugs increasing potassium level in blood (potassium drugs, kaliysberegayushchy diuretics, inhibitors of angiotensin-converting enzyme).
Special instructions
At intravenous use of not divorced drug it need to be entered slowly.
With care: hyperpotassemia, renal failure, disturbances of a warm rhythm, oligouriya, anury, fluid lungs, profound heart failure.
Pregnancy and the period of a lactation
Use of drug for treatment of pregnant women and the feeding women perhaps on doctor's orders if the expected advantage exceeds risk for a fruit and the child.
Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
the Overdose
Symptoms - strengthening of side effects does not influence.
Symptomatic treatment.
The form of release and packing
On 2 ml or 5 ml of drug place in ampoules of steady orange glass of I type neutral hydrolytic.
On 5 ampoules put in blister strip packaging.
On 5 planimetric packs together with the instruction for medical use in the state and Russian languages place in a pack from cardboard (for 2 ml).
On 1 planimetric packing together with the instruction for medical use in the state and Russian languages place in a pack from cardboard (for 5 ml).
To Store storage conditions at
a temperature not higher than 25 S. Hranit out of children's reach!
Not to apply a period of storage of 5 years after an expiration date.
Prescription status
According to the prescription
the Producer
of Legasi Pharmasyyutikals Switzerland Ryurbergstrasse
21, 4127 GmbH Birsfelden, Switzerland
the Owner of the registration certificate
of Pharmasyyutikals HONEY Switzerland GmbH, Switzerland
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products
Representation of Pharmasyyutikalz HONEY Switzerland GmbH in the Republic of Kazakhstan: Almaty, Dostyk Ave. 97, office 8, ph. +7 727 264-17-94, fax: + 7 727 264-17-71
E-mail address: info.safety@meda-cis.com