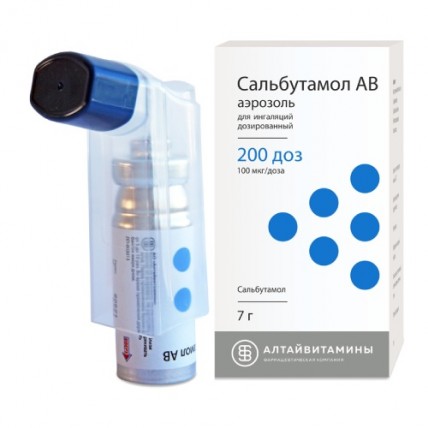
Salbutamol AV Inhaler 100 mcg/dose, 200 doses
- $15.00
What is Salbutamol?
Salbutamol is a medication that belongs to a class of drugs known as beta-2 agonists.
It is primarily used as a bronchodilator, which means it helps to widen the airways in the lungs, making it easier to breathe.
Salbutamol is commonly prescribed for people with respiratory conditions such as asthma, chronic obstructive pulmonary disease (COPD), and other disorders where breathing is impaired due to narrowing of the airways.
This medication is particularly effective in treating acute symptoms, such as sudden asthma attacks, where quick relief is essential.
Salbutamol is also used as a preventative measure to avoid symptoms triggered by exercise or allergens.
It can be found in various forms, including inhalers, tablets, and syrups, but the inhaler form is the most commonly used due to its rapid action and direct delivery to the lungs.
How Does a Salbutamol Inhaler Work?
A Salbutamol inhaler is designed to deliver quick relief directly to the lungs during an asthma attack or when experiencing breathing difficulties. Salbutamol is a medication that belongs to a group of drugs known as beta-2 agonists, which work specifically on the lungs.
When you use the inhaler, it releases a measured dose of Salbutamol sulfate, the active ingredient, into your airways. This medication works by binding to beta-2 receptors on the smooth muscles that surround the airways. Once bound, it causes these muscles to relax, which leads to the airways opening up, making it easier to breathe.
The inhaler provides fast-acting relief, typically within 5 minutes, which is why it is often referred to as a "rescue inhaler." The effects peak within 30 to 90 minutes and can last for 3 to 6 hours. This rapid action makes it highly effective for quickly stopping symptoms like wheezing, coughing, and shortness of breath.
In addition to its primary role of opening the airways, Salbutamol can also slightly increase heart rate and affect blood pressure, though these effects are usually minimal compared to its lung-specific actions.
By delivering the medication directly to the lungs, a Salbutamol inhaler ensures that most of the dose reaches the targeted area quickly, providing rapid symptom relief when it’s most needed. This direct and effective delivery system is why Salbutamol inhalers are a crucial part of managing asthma and other similar respiratory conditions.
What are the Benefits of Using a Salbutamol Inhaler?
The primary benefit of using a Salbutamol inhaler is its ability to provide quick relief during an asthma attack or other respiratory distress.
It is an essential part of managing chronic respiratory conditions, preventing episodes of breathlessness triggered by allergens, exercise, or other factors.
Moreover, it helps improve lung capacity and facilitates better mucus clearance in chronic conditions like bronchitis.
Who Can Use a Salbutamol Inhaler?
Salbutamol inhalers are suitable for adults and children over the age of 2.
However, the dosage and frequency must be carefully followed as prescribed by a healthcare professional.
It is commonly used by individuals with asthma, COPD, and other conditions that involve reversible airway obstruction.
How to Use a Salbutamol Inhaler?
- Preparation: Shake the inhaler well before use and remove the protective cap.
- Positioning: Sit or stand upright to ensure effective inhalation.
- Inhalation: Exhale fully, then place the mouthpiece in your mouth, ensuring a tight seal with your lips. As you start to inhale slowly, press the inhaler to release the medication.
- Breath-hold: After inhaling the medication, hold your breath for about 10 seconds to allow the medication to settle in your lungs.
- Repeat if necessary: If a second dose is needed, wait a few minutes and repeat the process.
Are There Any Side Effects?
Like all medications, Salbutamol inhalers can cause side effects, although not everyone experiences them.
Common side effects include a mild tremor, headache, or an increased heart rate.
Less commonly, it may cause muscle cramps, irritation of the throat, or paradoxical bronchospasm, which is a rare but serious reaction that requires immediate medical attention.
Can Pregnant Women Use a Salbutamol Inhaler?
Salbutamol should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
It is essential to consult with a healthcare provider before using Salbutamol during pregnancy or while breastfeeding.
How Should a Salbutamol Inhaler be Stored?
Store your Salbutamol inhaler at a temperature not exceeding 30°C (86°F) and keep it out of reach of children.
Avoid exposing the inhaler to direct sunlight or freezing temperatures, and do not puncture or burn the canister even when it appears empty.
When Should You Consult a Doctor?
If you find that your usual dose of Salbutamol is not providing the same relief or you need to use the inhaler more frequently than prescribed, it may indicate that your condition is worsening.
In such cases, consult your healthcare provider to reassess your treatment plan.