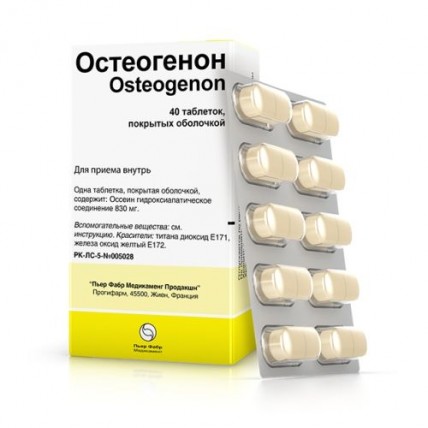
Osteogenon 830 mg, 40 film-coated tablets
- $65.00
Out Of Stock
Sku:
3fc7e2a948dd
Brand:
Pierre Fabre (France)
One tablet contains
Studies conducted on humans using isotopes (Ca47) have shown excellent absorption of the drug in the intestine. In comparison with simple calcium salts, Osteogenon did not create peaks of hypercalcemia; this is due to the slower absorption of ossein - hydroxyapatite complex in the intestine, as well as its continuous absorption.
For this reason, slow dissolution of the complex leads to better distal calcium absorption.
Osteogenon contains organic components of bone tissue (ossein) and a mineral bone component (microcrystalline hydroxyapatite), consisting of calcium and phosphorus in a naturally balanced ratio.
Ossein contains collagen and non-collagen peptides that have a positive effect on bone formation.
Osteogenon is prescribed for adults.
- with osteoporosis: 2-4 tablets 2 times a day;
- when correcting the calcium-phosphorus balance during pregnancy and breastfeeding: 1-2 tablets per day;
- concomitant therapy of fractures in adults: 2 tablets 3 times a day;
The tablets are swallowed with a small amount of water.
The duration of the course of treatment depends on the diagnosis and is determined by the doctor.
Like all medicines, Osteogenon can cause side effects, although not everybody gets them.
The following side effects have been found, although the exact frequency of occurrence cannot be determined and, therefore, they are classified as with an unknown frequency of manifestations:
- long-term treatment with high doses of the drug can increase the concentration of calcium in the blood or urine (hypercalcemia and hypercalciuria)
- abdominal pain, constipation, nausea
- itching, rash
- hypersensitivity to the components of the drug
- hypercalcemia, hypercalciuria
- calcium lithiasis
- prolonged immobilization
- children's age up to 18 years
- Patients with severe renal failure and patients on hemodialysis
With the concomitant administration of certain tetracycline antibiotics or iron-containing drugs, Osteogenon may reduce their absorption. For this reason, you should not take the above drugs at the same time and the interval between taking these drugs and Osteogenon should be at least 4 hours.
Joint reception with cardiac glycosides causes cardioarrhythmia. The interval between doses of drugs should be at least 4 hours.
Co-administration with bisphosphonates - there may be a risk of reduced intestinal absorption of the latter. It is recommended to take Osteogenon 4 hours after taking bisphosphonates.
Thiazide diuretics - there is a risk of developing hypercalcemia as a result of a decrease in calcium excretion in the urine.
When Osteogenon, calcium and vitamin D are combined, patients should be monitored for hypercalcemia and hypercalciuria.
In patients with high concentrations of calcium in the blood and urine, the dosing regimen should be adjusted.
In patients at high risk of urolithiasis, as well as in patients suffering from renal disease, dose adjustment is necessary and long-term use of Osteogenon should be avoided.
In patients suffering from renal insufficiency, it is necessary to regularly monitor the level of calcium in the blood and urine, in addition, high doses of Osteogenon should be avoided.
In case of long-term therapy and/or renal disorders, it is necessary to check the level of calcium in the blood and if it exceeds 7.5 mmol/24 hours (300 mg/24 hours) in adults and from 0.12 to 0.15 mmol/24 hours in children, the dose should be reduced drug or temporarily stop treatment.
In patients with moderate renal impairment, monitoring for serum phosphorus is recommended.
Pregnancy and lactation
The results of clinical studies conducted with the participation of pregnant women did not show any risk to the fetus. The drug can be used during pregnancy and during breastfeeding after consulting a doctor.
Features of influence on the ability to drive vehicles and potentially dangerous mechanisms
The drug does not affect the ability to drive vehicles and work with mechanisms.
Symptoms - nausea, vomiting, polyuria, thirst, dehydration, arterial hypertension, vasomotor disorders, constipation.
Treatment - it is necessary to stop taking all calcium-containing drugs, including vitamin D. Depending on the stage of intoxication, rehydration therapy (restoration of water-salt balance), diuretics, corticosteroids, calcitonin, possibly in combination with peritoneal dialysis, are prescribed.
Store at a temperature not exceeding 30 ºС.
Keep out of the reach of children!
Shelf life - 4 years
Do not use after the expiration date.
- Active ingredient: ossein-hydroxyapatite compound 830 mg (corresponding to non-collagen proteins/peptides 75 mg, collagen proteins/peptides 216 mg, calcium 178 mg and phosphorus 82 mg)
- Excipients: potato starch, microcrystalline cellulose, colloidal silicon dioxide, magnesium stearate
- Shell composition: hydroxypropyl methylcellulose, polyethylene glycol, talc, titanium dioxide E171, iron (III) oxide yellow E172
Pharmacological properties
Pharmacokinetics
Studies conducted on humans using isotopes (Ca47) have shown excellent absorption of the drug in the intestine. In comparison with simple calcium salts, Osteogenon did not create peaks of hypercalcemia; this is due to the slower absorption of ossein - hydroxyapatite complex in the intestine, as well as its continuous absorption.
For this reason, slow dissolution of the complex leads to better distal calcium absorption.
Pharmacodynamics
Osteogenon contains organic components of bone tissue (ossein) and a mineral bone component (microcrystalline hydroxyapatite), consisting of calcium and phosphorus in a naturally balanced ratio.
Ossein contains collagen and non-collagen peptides that have a positive effect on bone formation.
Indications for use
- Osteoporosis of various origins
- Accelerated healing of bone fractures
- Correction of osteopenia and regulation of calcium-phosphorus balance during pregnancy and lactation
Dosage and administration
Osteogenon is prescribed for adults.
- with osteoporosis: 2-4 tablets 2 times a day;
- when correcting the calcium-phosphorus balance during pregnancy and breastfeeding: 1-2 tablets per day;
- concomitant therapy of fractures in adults: 2 tablets 3 times a day;
The tablets are swallowed with a small amount of water.
The duration of the course of treatment depends on the diagnosis and is determined by the doctor.
Side effects
Like all medicines, Osteogenon can cause side effects, although not everybody gets them.
The following side effects have been found, although the exact frequency of occurrence cannot be determined and, therefore, they are classified as with an unknown frequency of manifestations:
- long-term treatment with high doses of the drug can increase the concentration of calcium in the blood or urine (hypercalcemia and hypercalciuria)
- abdominal pain, constipation, nausea
- itching, rash
Contraindications
- hypersensitivity to the components of the drug
- hypercalcemia, hypercalciuria
- calcium lithiasis
- prolonged immobilization
- children's age up to 18 years
- Patients with severe renal failure and patients on hemodialysis
Drug Interactions
With the concomitant administration of certain tetracycline antibiotics or iron-containing drugs, Osteogenon may reduce their absorption. For this reason, you should not take the above drugs at the same time and the interval between taking these drugs and Osteogenon should be at least 4 hours.
Joint reception with cardiac glycosides causes cardioarrhythmia. The interval between doses of drugs should be at least 4 hours.
Co-administration with bisphosphonates - there may be a risk of reduced intestinal absorption of the latter. It is recommended to take Osteogenon 4 hours after taking bisphosphonates.
Thiazide diuretics - there is a risk of developing hypercalcemia as a result of a decrease in calcium excretion in the urine.
Special instructions
When Osteogenon, calcium and vitamin D are combined, patients should be monitored for hypercalcemia and hypercalciuria.
In patients with high concentrations of calcium in the blood and urine, the dosing regimen should be adjusted.
In patients at high risk of urolithiasis, as well as in patients suffering from renal disease, dose adjustment is necessary and long-term use of Osteogenon should be avoided.
In patients suffering from renal insufficiency, it is necessary to regularly monitor the level of calcium in the blood and urine, in addition, high doses of Osteogenon should be avoided.
In case of long-term therapy and/or renal disorders, it is necessary to check the level of calcium in the blood and if it exceeds 7.5 mmol/24 hours (300 mg/24 hours) in adults and from 0.12 to 0.15 mmol/24 hours in children, the dose should be reduced drug or temporarily stop treatment.
In patients with moderate renal impairment, monitoring for serum phosphorus is recommended.
Pregnancy and lactation
The results of clinical studies conducted with the participation of pregnant women did not show any risk to the fetus. The drug can be used during pregnancy and during breastfeeding after consulting a doctor.
Features of influence on the ability to drive vehicles and potentially dangerous mechanisms
The drug does not affect the ability to drive vehicles and work with mechanisms.
Overdose
Symptoms - nausea, vomiting, polyuria, thirst, dehydration, arterial hypertension, vasomotor disorders, constipation.
Treatment - it is necessary to stop taking all calcium-containing drugs, including vitamin D. Depending on the stage of intoxication, rehydration therapy (restoration of water-salt balance), diuretics, corticosteroids, calcitonin, possibly in combination with peritoneal dialysis, are prescribed.
Storage conditions
Store at a temperature not exceeding 30 ºС.
Keep out of the reach of children!
Shelf life - 4 years
Do not use after the expiration date.