Ceraxon® (Citicoline)
- $38.00
What is Ceraxon® (Citicoline)?
Ceraxon® is a neuroprotective and nootropic medication that contains citicoline, a naturally occurring compound that plays a key role in the health of brain cells.
Citicoline is essential for maintaining cell membrane integrity and supporting the production of neurotransmitters, which help improve cognitive functions such as memory, learning, and focus.
Ceraxon® is often used in the treatment of neurological conditions, especially those related to stroke recovery or traumatic brain injuries.
By promoting the regeneration of nerve cells and improving blood flow in the brain, it helps speed up recovery from brain damage and enhances overall brain health.
What are the forms and dosages of Ceraxon®?
Ceraxon® is available in multiple forms and dosages, each designed to suit different treatment needs:
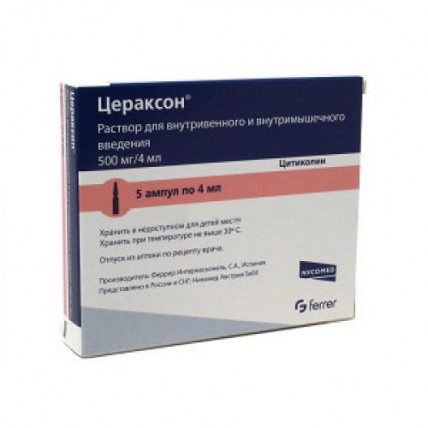
Injectable solution: Ceraxon® comes in ampoules of 500 mg/4 ml and 1000 mg/4 ml. This form is administered via intravenous (IV) or intramuscular (IM) injection. The injectable solution is typically used in hospital settings for more immediate or acute treatment needs. The IV solution is injected slowly over 3-5 minutes, or administered via drip at 40-60 drops per minute, depending on the dosage.
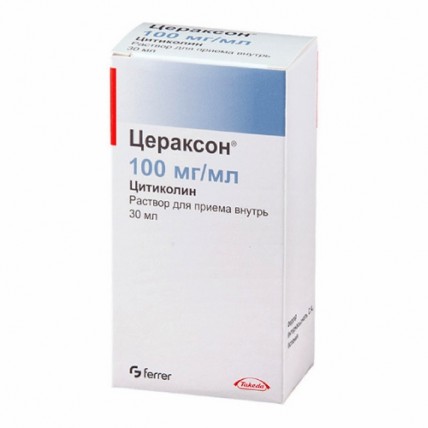
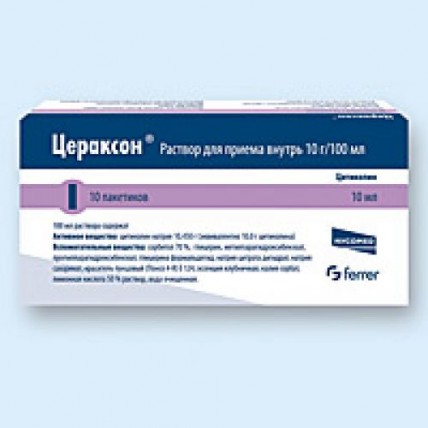
Oral solution (single-dose packets): A more convenient option for at-home use, the oral solution comes in 10 g/100 ml strength and is packaged in individual sachets for single-dose use. Each sachet typically contains 10 ml of solution, and it can be taken directly or diluted in water before consumption.
Oral solution (30 ml vial): This version is provided in a 30 ml vial, which includes a dosing syringe to ensure precise measurements. This form contains 10,450 mg of citicoline sodium per 100 ml, which is equivalent to 10,000 mg of citicoline. The solution can either be taken directly or mixed with a half-glass of water (120 ml). After use, the syringe should be rinsed with water.
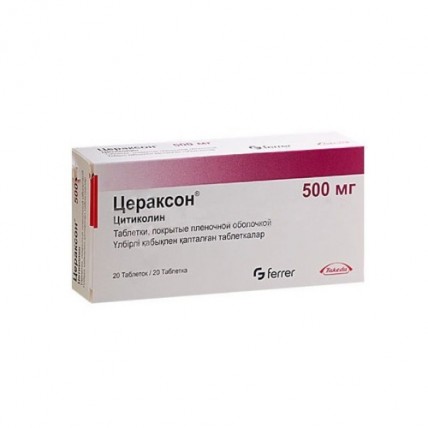
Tablets: Ceraxon® is also available in film-coated tablets, each containing 500 mg of citicoline. These are typically prescribed for long-term treatment and are easy to take, making them a preferred option for individuals managing chronic conditions.
Each form and dosage is chosen based on the patient's condition and severity of the disorder, with the injectable form used for acute cases and oral forms more suitable for long-term management.
What is Ceraxon® used for?
Ceraxon® is commonly prescribed for a variety of neurological conditions, especially those involving cognitive impairment or brain injuries.
Some of the primary uses include:
Neurological and cognitive disorders resulting from stroke: Ceraxon® plays a crucial role in aiding the recovery of brain functions impaired by a stroke. It helps to repair damaged brain cells and restore normal cognitive functions such as memory, focus, and problem-solving skills. By improving blood circulation and enhancing neural regeneration, Ceraxon® speeds up recovery and improves the quality of life for stroke survivors.
Treatment of traumatic brain injury (TBI): Ceraxon® is used to treat cognitive and neurological symptoms resulting from head injuries. These injuries can cause long-term damage to brain cells, and citicoline aids in the repair process, helping to restore mental clarity, focus, and motor skills. It also reduces the risk of further complications following a traumatic brain injury.
Cognitive decline related to degenerative or vascular brain conditions: Ceraxon® is often prescribed to individuals suffering from age-related cognitive decline, such as Alzheimer’s disease or dementia. It improves memory, attention, and overall brain function, especially in cases where cognitive issues are caused by decreased blood flow to the brain or other degenerative changes.
How effective is Ceraxon® in improving cognitive function compared to other nootropic supplements?
Ceraxon® (citicoline) has been shown to be highly effective in enhancing cognitive function, particularly in areas such as memory, focus, and mental clarity.
Citicoline works by boosting the production of essential neurotransmitters like acetylcholine, which plays a key role in memory and learning.
It also supports the regeneration of nerve cells and improves brain energy metabolism, which is crucial for maintaining mental sharpness.
Studies have found that citicoline can significantly improve cognitive performance, particularly in aging adults and those with mild cognitive impairment.
For example, it has been shown to enhance memory recall, increase attention span, and improve information processing speed.
Compared to other nootropic supplements, citicoline stands out due to its neuroprotective properties, helping to repair and maintain the structural integrity of brain cells.
Additionally, citicoline has demonstrated benefits in people recovering from neurological injuries, such as stroke or traumatic brain injury, further distinguishing it from other cognitive enhancers.
Its ability to improve cognitive function in both healthy individuals and those with neurological conditions makes it one of the more versatile and effective nootropic supplements available.
How should Ceraxon® be used?
The dosage and method of administration for Ceraxon® depend on the patient's condition, the severity of the disorder, and the form of the medication being used.
Injectable form: The recommended dosage ranges between 500 mg and 1000 mg per day, with higher doses used for more severe cases. The solution should be injected slowly (over 3 to 5 minutes), or via intravenous drip at 40-60 drops per minute. Once the ampoule is opened, the solution must be used immediately.
Oral solution (single-dose packets): Each sachet contains 10 ml of solution and can be taken directly or mixed with water. The contents of the sachet must be consumed immediately after opening, as the solution cannot be stored for later use.
Oral solution (30 ml vial): The 30 ml solution is administered using a dosing syringe for precision. The solution can be taken directly from the syringe or diluted in a half-glass of water (120 ml). After each dose, it is essential to clean the dosing syringe to maintain hygiene and ensure proper dosing. The recommended dose typically ranges from 500 mg to 2000 mg per day, depending on the severity of the condition. The minimum duration of treatment is 45 days, but longer treatment may be required in chronic conditions.
Tablets: The dosage for tablets typically ranges from 500 mg to 2000 mg per day (1-4 tablets daily), depending on the severity of the disease. Tablets should be taken as prescribed by a healthcare provider and should not be skipped or doubled up if a dose is missed.
Always follow the specific instructions given by your healthcare provider for the appropriate dosage and duration of treatment.
Who should not use Ceraxon®?
Ceraxon® is not suitable for everyone and should be avoided in the following cases:
Allergies or hypersensitivity: People who are allergic to citicoline or any of the other ingredients in Ceraxon® should avoid using the medication, as it may trigger allergic reactions such as rashes, swelling, or more severe symptoms like difficulty breathing.
Increased parasympathetic nervous system activity: This condition, which may manifest as low blood pressure, bradycardia, excessive sweating, and fainting, is a contraindication for Ceraxon®. In such cases, the drug could exacerbate the symptoms and worsen the patient’s condition.
In addition, patients with certain health conditions, particularly severe hypotension or bradycardia, should discuss their medical history with a healthcare professional before starting Ceraxon®.
Can Ceraxon® interact with other medications?
Yes, Ceraxon® can interact with certain medications.
It is important to inform your healthcare provider about all the medicines you are currently taking, as some interactions could affect your treatment outcome:
L-dopa: Ceraxon® has been shown to enhance the effects of L-dopa, a medication commonly used in the treatment of Parkinson’s disease. This interaction could potentially increase the therapeutic effect of L-dopa, but it may also increase the risk of side effects, such as dizziness or involuntary movements. Consult your doctor before combining these treatments.
Meclofenoxate: Ceraxon® should not be taken with drugs containing meclofenoxate, as both are psychostimulants. This could lead to overstimulation of the brain, increasing the risk of side effects such as insomnia, agitation, or headaches.
Always consult your doctor or pharmacist before starting any new medication to avoid harmful drug interactions.
Are there any special precautions when using Ceraxon®?
There are a few important precautions to be aware of when using Ceraxon®:
Fructose intolerance: The oral solution contains sorbitol, a type of sugar alcohol. If you have a hereditary condition known as fructose intolerance, you should consult your healthcare provider before taking this medication.
Pediatric use: There is limited data on the safety and efficacy of Ceraxon® in children. Therefore, it should only be prescribed to children when absolutely necessary, and the potential benefits must outweigh any risks.
Pregnancy and breastfeeding: The safety of citicoline during pregnancy or breastfeeding has not been fully established. Ceraxon® should only be used during pregnancy or lactation when clearly needed, and only if the potential benefits outweigh the risks to the mother or baby.
Storage instructions
Ceraxon® should be stored at a temperature between 15°C and 30°C (59°F and 86°F).
For the 30 ml oral solution, make sure the vial is kept out of direct sunlight and stored in a cool, dry place.
Like all medicines, Ceraxon® should be kept out of reach of children to prevent accidental ingestion.
Do not use the medication past the expiration date marked on the packaging.