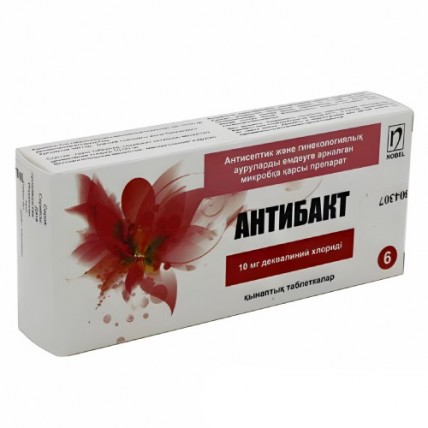
Antibact® (Dequalinium Chloride) 10 mg, 6 vaginal tablets
- $20.00
- $22.00
What is Antibact® (Dequalinium Chloride) 10 mg?
Antibact® is a vaginal tablet that contains 10 mg of Dequalinium Chloride, a medication specifically designed for the treatment of bacterial vaginosis.
This medicine belongs to a group of gynecological anti-infective and antiseptic agents.
It is primarily used to manage infections in the urogenital system, providing targeted relief and promoting vaginal health.
How does Antibact® (Dequalinium Chloride) work?
Antibact® works by utilizing the antimicrobial properties of Dequalinium Chloride, which effectively combats harmful bacteria that cause bacterial vaginosis.
By directly applying the tablet into the vagina, the active ingredient is delivered where it is most needed, reducing symptoms such as discharge, odor, and discomfort within a few days.
Who should not use Antibact® (Dequalinium Chloride)?
Antibact® should not be used by individuals who are hypersensitive to Dequalinium Chloride or any of the tablet’s other components.
It is also contraindicated for those with ulcers on the vagina or cervix, as well as in young girls who have not yet reached puberty.
Pregnant women should avoid using this medication within 12 hours of childbirth to minimize any potential impact on the newborn.
What precautions should be taken when using Antibact®?
Before using Antibact®, it is essential to consider the following precautions:
- Avoid using soaps or vaginal douches: Anions, such as those found in soaps, spermicides, and vaginal douches, can reduce the antimicrobial effectiveness of Dequalinium Chloride.
- During pregnancy and breastfeeding: Antibact® should only be used during pregnancy if absolutely necessary and not within 12 hours before delivery. It is generally safe for breastfeeding mothers as the systemic absorption is minimal, but should be used only if clinically justified.
- Consult a doctor if symptoms persist: If symptoms do not improve after the treatment course or if there is a recurrence, consult a healthcare provider for further advice.
How should Antibact® (Dequalinium Chloride) be used?
Antibact® is designed for intravaginal use. The recommended dosage is one vaginal tablet (10 mg of Dequalinium Chloride) per day for six days.
The tablet should be inserted deep into the vagina at night, preferably while lying on your back with your legs slightly bent.
If you are menstruating, pause the treatment and resume it after your period ends.
What should be done in case of an overdose?
While there are no specific symptoms associated with an overdose of Antibact®, using higher than recommended doses may lead to vaginal ulcerations.
If an overdose is suspected, discontinue use and seek immediate medical attention. Do not double the dose if a tablet is missed.
What are the possible side effects of Antibact®?
Common side effects may include:
- Vaginal discharge, itching, or burning
- Vaginal candidiasis (yeast infection)
Less common side effects can include:
- Vaginal bleeding or pain
- Vulvovaginitis (inflammation of the vulva and vagina)
- Headaches or nausea
In rare cases, ulceration of the vaginal lining, allergic reactions, or cystitis may occur. If you experience any severe or persistent side effects, discontinue use and seek medical help immediately.
How should Antibact® (Dequalinium Chloride) be stored?
Store Antibact® tablets at a temperature not exceeding 25°C (77°F) in a dry, well-protected place away from light and out of reach of children.
Do not use the tablets after the expiration date indicated on the package, which is two years from the date of manufacture.