Description
The instruction for medical use
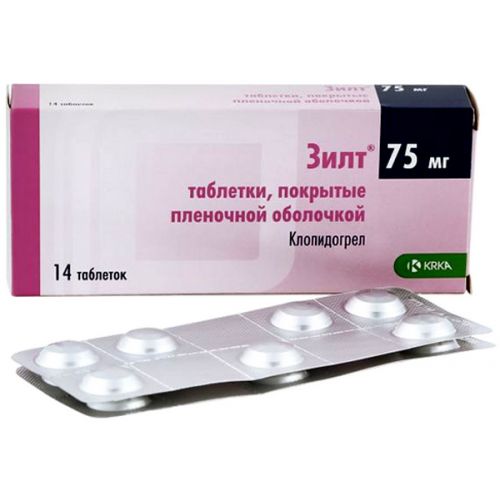
of ZILT® medicine
Trade name
ЗИЛТ®
the International unlicensed
name Klopidogrel Lekarstvennaya
the Tablet form, film coated, 75 mg
Structure
One tablet contains
active agent – klopidogrelya hydrosulphate of 97.875 mg 75 mg klopidogrelya,
excipients are equivalent: lactose anhydrous, cellulose mikrokristal-
lichesky, starch prezhelatinizirovanny, a macrogoal 6000, the castor oil
hydrogenated
structure of a cover: gipromelloza, titan dioxide (E171), ferrous oxide kras-
ny (E172), talc, propylene glycol.
Description
Biconvex round tablets, film coated, pink color
Pharmacotherapeutic group
Anticoagulants. Inhibitors of aggregation of thrombocytes.
The code of automatic telephone exchange B01AC04
the Pharmacological
Pharmacokinetics Later properties of oral administration klopidogrel is very quickly absorbed (tmax = 0.7 – 0.8 hours). 2 hours later in serum it is impossible to find even its traces. Irrespective of meal 50% of drug are soaked up. The most part klopidogrelya and its main metabolites (98% and 94% respectively) reversibly contacts plasma proteins.
Klopidogrel is pro-medicine. He is metabolized in a liver. The main metabolite – carboxyl derivative – is pharmacological not active. It makes about 85% of the substance circulating in plasma. It reaches the maximum serumal concentration approximately in 1 hour after use. The active metabolite – thiol derivative – is formed at oxidation klopidogrelya up to 2-oxo-klopidogrelya and the subsequent hydrolysis. Oxidation is provided with the system of P450 cytochrome (isoenzymes 2B6 and 3A4 and to a lesser extent also 1A1, 1A2 and 2C19). An active metabolite it is irreversible contacts receptors of thrombocytes and, thus, inhibits their aggregation. Still it was not isolated from serum yet.
After the single and repeating use of 50% klopidogrelya 46% with a stake are removed with kidneys, and. Time of semi-elimination of the main metabolite after the single and repeating use is 8 hours.
After use klopidogrelya at patients with a heavy renal failure (clearance of creatinine of 0.08-0.25 ml / with and/or 5-15 ml/min.) concentration of the main metabolite klopidogrelya was much lower, than at patients with a moderate renal failure (clearance of creatinine 0.5-1 of ml / with and/or 30-60 ml/min.). The inhibition of aggregation of thrombocytes was less, than at healthy faces (approximately only for 25%), the bleeding time was not extended in comparison with healthy subjects.
The pharmacodynamics
of Zilt® prevents linking of adenosinediphosphate (ADF) with the receptors located on a surface of thrombocytes and activation of GP IIb/IIIa of a complex. It leads to inhibition ADF-zavisimoy of aggregation of thrombocytes. Also it leads to inhibition of aggregation of thrombocytes under the influence of other incentives. The effect of ADF produced in each activated thrombocyte decreases and aggregation of the remained thrombocytes is stimulated. The effect klopidogrelya lasts during all life of a thrombocyte, platelet function is restored in 7-10 days after drug withdrawal of Zilt®.
At long purpose of therapeutic doses of the drug Zilt® (75 mg a day) the noticeable inhibition of aggregation of thrombocytes is noted in the first day of treatment, then the antiaggregantny effect gradually increases and reaches a maximum for the 3-7th day of regular administration of drug. At long reception of therapeutic doses the average level of inhibition is between 40 and 60%. After the end of treatment the effect klopidogrelya on aggregation and a bleeding time usually decreases within 5 days.
Indications
– prevention of aterotrombotichesky disturbances at the patients with
a myocardial infarction (to 35 days after a myocardial infarction) who had an ischemic
stroke (from 7 days to 6 months) or with the established disease
of peripheric circulation with occlusion elements
– the patients suffering from a syndrome of acute coronary insufficiency:
1) the syndrome of acute coronary insufficiency without increase
ST segment (unstable stenocardia or a myocardial infarction
without tooth Q), including patients with cherezkozhny coronary
placement of a stent (in a combination with acetilsalicylic
acid)
2) an acute myocardial infarction with increase in a segment of ST (in
to a combination with acetylsalicylic acid at patients,
which showed thrombolytic therapy)
the Route of administration and doses
the Recommended drug Zilt® dose for adults (including elderly patients) makes 1 tablet (75 mg) a day irrespective of meal.
Duration of a course of treatment is determined by the attending physician.
At the patients suffering from a syndrome of acute coronary insufficiency:
1) a syndrome of acute coronary insufficiency without increase in a segment of ST (unstable stenocardia or a myocardial infarction without tooth Q), treatment has to be begun with a single introduction dose of 300 mg, and then is continued by a dose of 75 mg once a day (with acetylsalicylic acid in a dose of 75 – 325 mg a day). As higher doses of acetylsalicylic acid are accompanied by the increased risk of bleeding, it is not recommended to exceed a dose of acetylsalicylic acid of 100 mg. The optimum duration of treatment is formally not established. Data of clinical trials confirm use of the scheme during up to 12 months, and the maximum advantage is observed after 3 months,
2) an acute myocardial infarction with increase in a segment of ST: Зилт® it can be applied in the form of a single daily dose of 75 mg, with an initiation of treatment an introduction dose in a combination with acetylsalicylic acid with other trombolitika or without them. At patients 75 years are more senior treatment klopidogrely should be begun without introduction dose. Combination therapy is begun as soon as possible after emergence of symptoms and continued within, at least, four weeks.
There are no data on studying advantages of use over 4 weeks of combination therapy by the drug Zilt® with acetylsalicylic acid.
Side effects
– a stethalgia, grippopodobny symptoms, pain, fatigue, an asthenia
– a headache, dizziness, paresthesia, spasms of legs, a hyperesthesia,
neuralgia
– a syncope, heartbeat, peripheral hypostases, arterial hypertension,
generalized hypostases, heart failure
– nausea, vomiting, an abdominal pain, dyspepsia, diarrhea, a constipation, a meteorism,
perforation of stomach ulcer
– syp, an itching
– a purpura, epistaxis, of bleeding gastrointestinal, vnutrisustav-
ny, bleeding from urinary tract, a pneumorrhagia,
intracraneal hemorrhages, retroperitoneal hemorrhages,
bleedings from postoperative wounds, intraocular hemorrhages,
a hemothorax, pulmonary bleedings
– a neutropenia, thrombocytopenia, a leukopenia, an eosinophilia
Very seldom
– a Werlhof’s trombogemolitichesky disease, heavy
thrombocytopenia, an agranulocytosis, a granulocytopenia, anemia aplastic
or hypochromia, leukemia
– anaphylactoid reactions, a serum disease
– confusion of consciousness, a hallucination, a depression
– flavoring disturbances
– a vasculitis, hypotension
– cough, rhinitis, a bronchospasm, bronchitis, an interstitial pneumonitis,
pneumonia, sinusitis
– pancreatitis, colitis (ulcer or lymphocytic)
– stomatitis
– an acute liver failure, hepatitis
– a Quincke’s disease, bullous rash (mnogoformny erythema,
Stephens-Johnson’s syndrome, a toxic necrolysis of epidermis), erythematic
rash, a small tortoiseshell, eczema and flat deprive
– arthralgias, arthritises, arthroses, myalgias
– cystitis, a glomerulonephritis
– menorrhagias
– temperature increase
– an abnormal liver function, increase in creatinine of blood, giperbiliru-
a binemiya
Isolated cases
– a hemolytic uraemic syndrome, a hymenoid nephropathy
of the Contraindication
– hypersensitivity to klopidogrelyu and/or to any of
drug components
– the profound liver failure
– a hemorrhagic syndrome, acute bleeding (including intracraneal
hemorrhage) and the diseases contributing to its development
(a peptic ulcer of a stomach and a 12-perstny gut in aggravation stages,
nonspecific ulcer colitis, tuberculosis, tumors of lungs,
a hyper fibrinolysis)
– pregnancy and the period of a lactation
– the children’s and teenage age up to 18 years (the efficiency and safety
of medicine is not established)
Medicinal interactions
is not recommended co-administration of Zilt® and warfarin in connection with the increased risk of bleeding.
Зилт® it is necessary to apply with care at the patients subject to the increased risk of bleeding connected with an injury, surgical interventions or other morbid conditions, receiving IIb/IIIa glycoprotein inhibitors.
Acetylsalicylic acid does not change the adenozindifosfatindutsirovan-ny aggregation of thrombocytes caused klopidogrely. Periodic prescribing of acetylsalicylic acid, 500 mg 2 times a day, did not cause lengthening of a bleeding time. Зилт® can exponentiate effect of acetylsalicylic acid on the aggregation of thrombocytes induced by collagen, possible pharmakodinamichesky interaction klopidogrelya and acetylsalicylic acid, increases risk of bleeding therefore combined use the drug Zilt® and acetylsalicylic acid possible no more than one year. The question of combined use of these drugs has to be comprehensively estimated.
Due to the increased risk of bleeding it is recommended to be careful at co-administration of the drug Zilt® with heparin, thrombolytic drugs.
Co-administration of the drug Zilt® and non-steroidal anti-inflammatory drugs increases risk of developing ulcers of digestive tract and ulcer bleedings. Therefore it is necessary to be careful at their simultaneous appointment.
Significant clinical interactions at the co-administration of the drug Zilt® and atenolol, AKF inhibitors, means reducing cholesterol content in blood of nifedipine, digoxin, phenobarbital, Cimetidinum, estrogen, or theophylline are not revealed.
Зилт® inhibits activity of CYP2C9 enzyme of a system of P450 cytochrome at simultaneous use with the drugs which are metabolized with the participation of this enzyme (Phenytoinum, tolbutamide) the increase in their concentration in blood plasma is possible.
Antacids do not influence absorption klopidogrelya.
Except for specific information on the medicinal incompatibility given above the tests on interaction of the drug Zilt® and some medicines which are usually applied at patients aterotrombozy were not carried out. However, the patients participating in clinical trials klopidogrelya received along with it various drugs, including diuretics, blockers of beta receptors, AKF inhibitors, antagonists of calcium channels, the drugs reducing cholesterol level in blood, coronary vazodilyator, anti-diabetic drugs (including insulin), anti-epileptic means, gormonozamestitelny therapy and antagonists of GPIIb/IIIa, without signs of clinical significant collateral interactions.
Special instructions
Due to the risk of bleeding and hematologic side effects, in case of emergence during treatment of the clinical symptoms indicating it it is necessary to carry out blood test immediately.
As well as to apply other antithrombocytic drugs, Zilt®sleduet with care at the patients subject to the increased risk of bleeding connected with an injury, surgical interventions or other morbid conditions and also in case of combination klopidogrelya with acetylsalicylic acid, non-steroidal anti-inflammatory drugs including TsOG-2 inhibitors, heparin, or IIb/IIIa glycoprotein inhibitors. It is necessary to control carefully patients on presence of symptoms of bleeding, including the concealed hemorrhages, especially within the first weeks of treatment and/or after the invasive warm procedures or surgical intervention.
Combined use of Zilt® with anticoagulants for oral administration is not recommended owing to a possibility of increase in intensity of bleedings.
The Zilt®dolzhno drug treatment to be stopped at least in 7 days prior to the planned surgery (including dental procedures).
Patients have to inform the doctor and the stomatologist on administration of drug if surgeries are coming them or if the doctor appoints medicine, new to the patient.
Зилт® extends a bleeding time. It has to be appointed with care to patients with the increased risk of bleeding after injuries, operations or as a result of other morbid conditions and also to patients with tendency to bleedings (especially gastrointestinal and intraocular hemorrhages).
Patients have to be warned that during the Zilt® drug treatment there is an increased tendency to bleeding and bleeding can last slightly longer, than usually before independently stopping. In case of small cuts (for example, when shaving) and injuries usually are not required any special measures. At big cuts or injuries the immediate consultation with the doctor is necessary.
Very seldom, after drug Zilt® use sometimes of short, cases of the trombotichesky Werlhof’s disease (TWD) were noted. They are characterized by the thrombocytopenia and mikroangiopatichesky hemolytic anemia interfaced to neurologic disturbances, dysfunction of kidneys or a hyperthermia. The TPP is the state demanding immediate intervention including a plasma exchange.
Owing to lack of data, reception of Zilt® should not begin in the first 7 days after an acute ischemic stroke.
Patients with impaired renal function should use drug with care.
Patients with a moderate liver failure need to apply with care Zilt® in a type of the increased risk of bleeding.
Зилт® contains lactose. To patients with hereditary intolerance of a galactose, with a galactosemia, deficiency of Lappa lactase or a sprue of glucose galactose it is not recommended to take this drug.
Зилт® contains the hydrogenated castor oil which can be the cause of indigestion and diarrheas.
The feature of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
during treatment needs to be careful during the driving of motor transport and occupation other potentially dangerous types of activity demanding the increased concentration of attention and speed of psychomotor reactions.
Overdose
Symptoms: lengthening of a bleeding time and subsequent complications.
Treatment is symptomatic. If fast correction of the extended bleeding time is necessary, action klopidogrelya can be suspended by transfusion of thrombocytes. Specific antidote is absent.
Form of release and packing
of the Tablet, film coated, 75 mg.
On 7 tablets in blister strip packaging from a film of polyvinylchloride and aluminum foil. On 2 or 4 packs together with the instruction for use in the state and Russian languages put in a cardboard pack.
To Store storage conditions at
a temperature not higher than 25 Store OS out of children’s reach!
A period of storage
3 years
not to apply after a period of storage
Prescription status
According to the prescription
KRKA-FARMA Producer, of the lake of the lake, Yastrebarsko’s DPC, Tsvetkovichi bb, Croatia to KRK, of, the place, Slovenia
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products (goods)
Representation “KRK, of Is new. The place” in PK
PK is new, 050059, Almaty, Al-Farabi Ave., 5/1, section 3 A, the 4th floor
ph.: +7 (727) 311 08 09
fax: +7 (727) 311 08 12
www.krka.si
to Develop
Additional information
| Ingredient |
|---|














Reviews
There are no reviews yet.