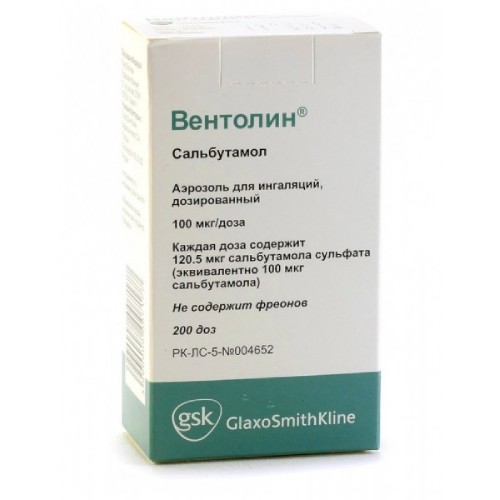
Description
Composition
One dose contains active ingredients – salbutamol sulfate 120.5 mcg (equivalent to salbutamol 100 mcg),
The excipient is 1,1,1,2-tetrafluoroethane (HFA-134a propellant), ozone-safe.
Pharmacological properties
Pharmacokinetics
Absorption
When using an aerosol, from 10 to 20% of the dose taken reaches the lower respiratory tract, where it is adsorbed by the lung tissue and penetrates into the vessels of the lungs, but is not metabolized here. The rest remains in the delivery device or settles in the oropharynx with further ingestion of the drug.
Distribution
Plasma protein binding is 10%.
Metabolism
Once circulatory thresholds are reached, salbutamol is metabolized via the hepatic mechanism and excreted predominantly in the urine as unchanged product and phenol sulfate.
Salbutamol is metabolized during the first passage through the liver and, due to the ingestion of small amounts during inhalation, in the intestinal wall; the main metabolite is an inactive sulfate conjugate, which is excreted in the urine.
breeding
T½ of salbutamol when administered intravenously is 4-6 hours. Salbutamol is rapidly excreted in the urine as an inactive 4′-O-sulfate metabolite and unchanged substance; in small amounts excreted with feces. Most of the salbutamol taken is eliminated from the body within 72 hours.
Pharmacodynamics
Ventolin® is a selective β2-adrenergic receptor agonist. In therapeutic doses, it affects the β2-adrenergic receptors of the bronchial muscles.
Ventolin® has a short duration of action (4 to 6 hours) and a fast onset of action (about 5 minutes from application).
Children
Clinical studies conducted in children under 4 years of age have proven a similar safety profile compared with older children, adolescents and adults.
Indications for use
As part of complex therapy:
– to stop and prevent the development of bronchospasm in patients with reversible airway obstruction (asthma, chronic bronchitis, emphysema)
– for stopping and preventing attacks of bronchial asthma before an expected attack
Bronchodilators should not be the sole or main component of asthma therapy. If a patient with asthma
does not respond to salbutamol therapy, it is recommended to use
inhaled corticosteroids to achieve and maintain symptom control. Insufficient response to salbutamol therapy may be a signal for urgent medical intervention/therapy.
Dosage and administration
Ventolin® in the form of an aerosol is used only for inhalation by inhalation of an aerosol through the mouth.
An increase in the need for taking β2-agonists may indicate a worsening of asthma. In such cases, it is recommended to re-evaluate the ongoing therapy and consider the possibility of additional administration of corticosteroids.
Due to the risk of adverse reactions when the recommended doses are exceeded, the frequency of administration and the doses used should be increased only as directed by a physician.
The duration of action of the drug Ventolin® in most patients is 4-6 hours.
Individuals who have difficulty coordinating inhalation and drug release from an inhaler can use Ventolin® with a spacer device.
The need to take the drug should not exceed 4 times a day (800 mg). A sudden increase in the need for the drug indicates a worsening of asthma.
For younger children, it is better to use Ventolin® through a spacer.
Relief of an acute attack of bronchospasm
Adults: 100 micrograms to 200 micrograms of Ventolin once.
Children: 100 mcg once. If necessary, the dose can be increased to 200 mcg.
Prevention of exercise-induced bronchospasm or allergic etiology
Adults: 200 mcg prior to exercise or anticipated allergen exposure
Children: 100 micrograms before exercise or expected allergen exposure. If necessary, the dose can be increased to 200 mcg.
Long-term maintenance therapy
Adults and children 100-200 mcg of the drug 4 times a day.
The duration of treatment is determined by the attending physician.
Instructions for using the inhaler
The effect of the drug may be weakened if the inhaler is cold. When cooling the can, it is recommended to remove it from the plastic case and warm it with your hands for several minutes. The can must not be disassembled, pierced or thrown into fire, even if it is empty.
Checking the health of the inhaler
Before using the inhaler for the first time, you must carefully remove the cap from the mouthpiece, shake the inhaler vigorously and spray two doses of the drug into the air to make sure that the device is working. If the inhaler has not been used for 5 days or more, shake it well and spray two doses of the drug into the air to make sure it is working.
Using an inhaler
1. Remove the protective cap from the mouthpiece of the inhaler. Check the inside and outside of the inhaler, including the mouthpiece, for cleanliness and dryness, as well as for loose parts of the device.
2. Shake the inhaler vigorously to mix the contents of the inhaler evenly and to remove all loose parts from the surface of the device.
3. Place the inhaler vertically between the thumb and forefinger, placing the thumb on the base, below the mouthpiece.
4. Take a deep (as far as possible) exhale. Then place the mouthpiece between your teeth (without biting it) and tightly clasp your lips.
5. Take a deep breath through your mouth. While continuing to take a deep breath, press the top of the inhaler.
6. Hold your breath, remove the inhaler from your mouth and remove your index finger from the top of the inhaler. Continue to hold your breath as much as possible.
7. If it is necessary to continue inhalation, wait approximately half a minute, holding the inhaler vertically, and then repeat steps 2 to 6.
8. After inhalation, carefully put the dust cap on the mouthpiece.
Attention
Take your time when performing the steps indicated in points 4, 5 and 6. It is important that the inhaler is pressed at the beginning of a calm, deep breath. To be sure that the inhalation is performed correctly, you must first control the method of taking the medicine in front of the mirror. The “haze” that appears during inhalation from the inhaler, lips or nose indicates an incorrect inhalation technique and it is necessary to practice using the inhaler again, starting from point 2.
If your doctor has given you any other advice on the use of the drug, follow your doctor’s recommendations. Inform your doctor about any difficulties associated with taking the drug.
Cleaning the inhaler
The inhaler should be cleaned at least once a week.
1. Remove the metal container from the plastic body of the inhaler and remove the mouthpiece cover.
2. Rinse the atomizer with warm running water.
3. Dry the atomizer thoroughly inside and out.
4. Replace the container and the mouthpiece cover.
DO NOT DIP THE METAL CONTAINER INTO WATER.
Side effects
Very often (>1/10), often (>1/100, <1/10), infrequently (>1/1,000, <1/100), rarely (>1/10,000, <1/1,000), very rarely (<1/10,000).
Often
– tremor, headache
– tachycardia
Infrequently
– irritation of the mucous membrane of the oral cavity and pharynx
– cardiopalmus
– muscle cramps
Seldom
– hypokalemia (beta2-agonist therapy can lead to severe hypokalemia)
– expansion of peripheral vessels
Very rarely
– hypersensitivity reactions, including urticaria, angioedema, bronchospasm, hypotension, collapse
– paradoxical bronchospasm
– lactic acidosis (in patients receiving salbutamol in the form of intravenous injections and through a nebulizer for the treatment of exacerbations of bronchial asthma)
– hyperactivity
– arrhythmia, including atrial fibrillation, supraventricular tachycardia and extrasystoles
Contraindications
– hypersensitivity to any component that is part of drug
– premature birth
– threatened abortion
Forms of release of salbumamol, not intended for intravenous administration, should not be used to stop preterm labor and threatened miscarriage.
Drug Interactions
It is not recommended to simultaneously use the drug Ventolin® and non-selective blockers of β-adrenergic receptors, such as propranolol.
Ventolin® is not contraindicated in patients receiving monoamine oxidase inhibitors (MAOIs).
Special instructions
Asthma is usually treated in stages, with the patient’s response monitored clinically and with lung function tests.
An increased need for β2-agonists may indicate worsening asthma control. In such cases, the patient’s treatment plan should be reviewed.
Sudden and progressive worsening of bronchial asthma can pose a threat to the life of the patient, therefore, in such situations, it is necessary to urgently address the issue of prescribing or increasing the dose of glucocorticosteroids. In such patients, daily monitoring of peak expiratory flow is recommended.
The drug is used with caution in patients with thyrotoxicosis.
Therapy with b2-adrenergic agonists, especially when administered parenterally or by nebulizer, can lead to hypokalemia. Particular caution is recommended in the treatment of severe asthma attacks, since in these cases hypokalemia may increase as a result of the simultaneous use of xanthine derivatives, glucocorticosteroids, diuretics, as well as due to hypoxia. In such situations, it is necessary to control the level of potassium in the blood serum. As with the use of other inhaled drugs, paradoxical bronchospasm may develop as a result of the onset of spasm immediately after a dose. If paradoxical bronchospasm occurs, immediate relief is required with an alternative drug or a fast-acting inhaled bronchodilator from another pharmacological group. You should immediately stop treatment with this form of Ventolin®, and, if necessary, prescribe other fast-acting bronchodilators for further use.
If the effect of the usual dose of the drug Ventolin becomes less effective or less prolonged (the effect of the drug should last at least 3 hours), the patient should consult a doctor.
The clinician must ensure that the patient is using the inhaler correctly and the timing between actuation of the device and inspiration for optimal drug delivery to the lungs.
Fertility
There is no data on the effect of the drug on fertility in humans. The drug does not adversely affect the fertility of animals.
Pregnancy and lactation
The use of the drug during pregnancy and lactation is justified only if the expected benefit to the mother outweighs the risk to the fetus/infant. Salbutamol is probably excreted in breast milk.
In separate studies, polydactyly and cleft palate have been identified in children with mothers taking drugs during pregnancy, among which was salbutamol (an unequivocal causal relationship of their occurrence with taking the drug has not been established), and therefore the risk is estimated at 2-3%. In experimental studies, the presence of a teratogenic effect of salbutamol was found: in mice with s / c administration (doses 11.5-115 times higher than the maximum recommended in humans for inhalation administration), the development of a “cleft palate” was noted; in rabbits, when administered orally (doses 2315 times higher than the maximum for inhalation administration), non-fusion of the skull bones.
Features of the influence of the drug on the ability to drive a vehicle or potentially dangerous mechanisms
No data.
Overdose
Symptoms: Most of the symptoms of an overdose of salbutamol are transient adverse reactions of beta-agonists.
In case of an overdose, hypokalemia may develop, and therefore it is necessary to monitor the level of potassium in the blood serum.
With the use of high therapeutic doses and an overdose of short-acting beta-agonists, the development of lactic acidosis was revealed.
Treatment: the use of large doses of salbutamol can cause hypokalemia, therefore, if an overdose is suspected, the level of potassium in the blood serum should be monitored. It is necessary to control the level of lactate and the subsequent development of metabolic acidosis (especially in the presence or worsening of tachypnea despite the elimination of bronchospasm).
Storage conditions
Store at a temperature not exceeding 30 ºС.
Protect from light and hypothermia.
Keep out of the reach of children!
Shelf life – 2 years
Do not use after the expiration date.
Additional information
| Ingredient |
|---|

















Reviews
There are no reviews yet.