Description
The instruction for medical use
of medicine
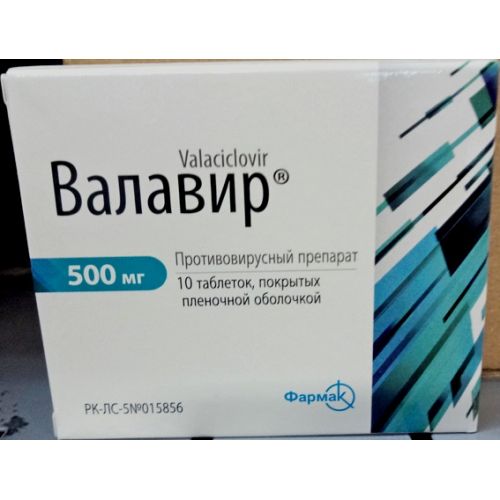
VALAVIR
the Trade name
Valavir
Mezhdunarodnoye the unlicensed
name Valatsiklovir Lekarstvennaya
the Tablet form, coated, 500 mg
Structure
One tablet contains
active agent – a valatsiklovir a hydrochloride (in terms of valatsiklovir 100% anhydrous substance) – 500 mg,
excipients: microcrystalline cellulose, polyvinylpirrolidone low-molecular medical, aerosil, krospovidon, magnesium stearate,
structure of a cover: Sepifilm 050: methylhydroxypropyl cellulose, cellulose microcrystalline, acetylated (or ethers of acetic acid) mono – and diglycerides, Kandurin: potassium-aluminum silicate (E 555), the titan dioxide (E 171)
the Description
of the Tablet of oblong shape with a biconvex surface, with risky, covered with a film nacreous cover, almost white color.
Pharmacotherapeutic group
Antiviral drugs of direct action. Nucleosides and nucleotides.
The automatic telephone exchange of J05A B11
The pharmacological
Pharmacokinetics Pharmacokinetics properties of a valatsiklovir and acyclovir is similar at single and repeated use. After intake valatsiklovir it is well soaked up, quickly and almost completely turns into acyclovir and valine under the influence of the enzyme of a valatsiklovirgidrolaza which is formed in a liver of the person. The bioavailability of acyclovir at reception of 1 g of a valatsiklovir is 54% and does not decrease at a concomitant use with food. The maximum concentration of acyclovir makes 10-37 µmol (2.2-8.3 mkg/ml) after single dose of 0.25-2 g by healthy volunteers with normal function of kidneys, and a median of time of achievement of this concentration 1-2 hours. The maximum concentration of a valatsiklovir in blood plasma is reached on average in 30–100 min. after administration of drug, by 3 h valatsiklovir in blood plasma is not defined. Linking of a valatsiklovir with proteins of blood plasma very low – 15%.
Valatsiklovir is brought with urine, mainly, in the form of acyclovir (more than 80% of a dose) and its metabolite a 9-karboksimetoksimetilguanin.
At patients with normal function of kidneys the elimination half-life of acyclovir makes 3 hours. At patients with an end-stage of a renal failure the elimination half-life of acyclovir makes about 14 hours.
The virus of shingles and a virus of a herpes simplex significantly do not change pharmacokinetics of a valatsiklovir after oral administration. In the last trimester of pregnancy the area under a curve acyclovir concentration/time in a plateau phase after use of a valatsiklovir in a dose of 1 g/days was approximately twice higher, than after acyclovir use orally in a dose of 1.2 g/days. At patients with HIV infection the pharmacokinetic characteristics of acyclovir after single or repeated use in a dose of 1-2 g of a valatsiklovir did not change in comparison with healthy faces.
At recipients of transplants of bodies which received valatsiklovir in a dose 2 g 4 times a day the maximum concentration of acyclovir was equal or exceeded concentration at healthy which received the same dose of drug, and daily indicators of the area under a curve concentration/time were much higher.
A pharmacodynamics
Valatsiklovir — antiviral drug, L-valinovy ether of acyclovir which is an analog of a purine (guanine) nucleoside. Is specific inhibitor of a DNA polymerase of viruses of herpes. Blocks synthesis of virus DNA and replication of viruses. In a human body valatsiklovir turns into acyclovir and valine. Acyclovir has specific activity concerning viruses of a herpes simplex (Herpes simplex virus-1) and II (Herpes simplex virus-2) of type, a virus of shingles (Varicella zoster virus), a cytomegalovirus (Cytomegalovirus), Epstein-Barr virus virus and a virus of herpes of the person of the VI type (Human herpesvirus-6). By phosphorylation, acyclovir turns into the active triphosphate of acyclovir competitively inhibiting synthesis of virus DNA. At the first stage of phosphorylation the activity of virus-specific enzyme is necessary. For a virus of a herpes simplex, a virus of shingles and Epstein-Burra’s virus it is the virus thymidinekinase which is available only in the cells infected with a virus. The partial selectivity of phosphorylation remains in a Cytomegaloviral infection and is mediated through UL97 phosphotransferase gene product. Acyclovir activation by specific virus enzyme considerably explains its selectivity.
Acyclovir phosphorylation process (transformation from mono – in triphosphate) is carried out by cellular kinases. Acyclovir triphosphate competitively inhibits a virus DNA polymerase and is built in virus DNA that leads to a complete separation of a chain, the termination of synthesis of DNA and blocking of replication of a virus.
The resistance is caused by deficit of a thymidinekinase of a virus that leads to excessive spread of a virus in the owner’s organism. Sometimes reduction of sensitivity to acyclovir is caused by emergence of virus strains from the virus thymidinekinase broken by structure or a DNA polymerase. At patients with the normal immune status the virus with reduced sensitivity to acyclovir occurs exclusively seldom and not often in patients with an immunodeficiency, for example, after bone marrow transplantation and other bodies, in the patients receiving chemotherapy in oncological diseases and at HIV-positive patients.
Valatsiklovir accelerates anesthesia at treatment of the surrounding herpes, reduces duration of a pain syndrome and also reduces the frequency of cases of patients with zosterassotsiirovanny pain, including with acute and post-herpetic neuralgia.
Prevention of a Cytomegaloviral infection by means of a valatsiklovir reduces risk of acute rejection of a transplant (patients after renal transplantation), the frequency of developing of the opportunistic infections and other infections caused by a virus of a herpes simplex and the Varicella zoster virus.
Indications
– treatment of the surrounding herpes (herpes zoster)
– treatment and suppression of a recurrence of infections of skin and the mucous membranes caused by a herpes simplex virus including primary and recurrent genital herpes
– treatment of labial herpes
– prevention of infection with genital herpes of the sexual partner as suppressive therapy (at observance of rules of safe sex)
– prevention of the diseases caused by a Cytomegaloviral infection after organ transplantation
the Route of administration and doses
Treatment of the surrounding herpes.
Appoint to adults and children (12 years are aged more senior) 1 g of Valavir 3 times a day. Course of reception of 7 days. Therapy needs to be begun as soon as possible at the first signs and symptoms, it is the most effective in the first 48 hours.
The infections caused by a herpes simplex virus.
In case of primary disease which can proceed more hard Valavir appoint to adults and children (12 years are aged more senior) 500 mg two times a day for 5 – 10 days.
For treatment of a recurrence Valavir appoint 500 mg 2 times a day for 3-5 days.
It is necessary to begin treatment as soon as possible, for recurrent forms of infections of a herpes simplex — during a prodromal stage or right after emergence of the first symptoms.
Preventive treatment (suppression) of a recurrence of the infections caused by a herpes simplex virus in adults and children (12 years are aged more senior).
The patient with the normal immune status appoint 500 mg of drug of 1 times a day.
The patient with very frequent aggravations (for example ≥10 episodes a year) the dose of 500 mg is appointed in 2 receptions (on 250 mg 2 times a day).
To patients with an immunodeficiency appoint 500 mg 2 times a day. Duration of treatment depends on risk of emergence of a recurrence of an infection and is established by the doctor individually.
For treatment of labial herpes Valavir’s use in a dose of 2 g 2 times a day with an interval of 12 hours is possible. Duration of a course of treatment should not exceed 1 day. Therapy should be begun at emergence of the first early symptoms (feelings of tingling, an itching or burning in lips).
For prevention of infection with genital herpes of the sexual partner as suppressive therapy.
To adult heterosexual persons with normal immunity at whom note 9 or less exacerbations of a disease within a year Valavir appoint 500 mg of 1 times a day within a year and more than an every day at regular sexual contacts, at irregular sexual contacts Valavir’s reception needs to be begun in 3 days prior to expected sexual contact.
Prevention and treatment of a Cytomegaloviral infection.
To adults and children (12 years are aged more senior) Valavir appoint 2 g 4 times a day as soon as possible after transplantation.
In a renal failure the dose is reduced. Treatment duration usually is 90 days, but can be increased at patients with a high risk.
The dosing mode at patients with a renal failure:
The therapeutic indication
Clearance of creatinine, ml/min.
Valavir
Herpes zoster
15-30
1’s Dose of 2 times in day
of ≤15
1 g of 1 times in day
of Herpes simplex (treatment)
of ≤15,500 mg of 1 times in day
of Herpes labialis (treatment)
of 31-49
1 g 2 times a day for 1 day
of 15-30,500 mg 2 times a day for 1 day
of ≤15,500 mg once
Herpes simplex (prevention)
• normal immunity
of ≤15,250 mg of 1 times a day
• an immunodeficiency
of ≤15,500 mg of 1 times a day
Reduction of risk of transfer of a virus in genital herpes
of ≤15,250 mg of 1 times a day
Prevention of a Cytomegaloviral infection:
Clearance of creatinine, ml/min.
Valavir’s Dose
of ≥75
2 g 4 times in day
of 50-75 1,5 g 4 times in day
of 25-50 1,5 g 3 times in day
of 10-25 1,5 g 2 times a day
≤10 or dialysis
of 1.5 g of 1 times a day
Side effects
Often
– a headache
– nausea
Seldom
– dizziness, disturbance and confusion of consciousness, a hallucination,
a loss of consciousness
– short wind
– discomfort in an abdominal cavity, vomiting, diarrhea
– skin rashes, including the photosensitization phenomena, an itching
– the renal failure
is Very rare
– a leukopenia, thrombocytopenia
– an anaphylaxis
– agitation, a tremor, an ataxy, a dysarthtia, psychotic symptoms,
convulsions, encephalopathy, a coma
– reversible increase in level of functional hepatic tests
– urticaria, a Quincke’s disease
– an acute renal failure
of the Contraindication
– hypersensitivity to a valatsiklovir, acyclovir or other
components of drug
– pregnancy and the period of a lactation
– children’s age up to 12 years
Medicinal interactions
of Signs of clinically significant medicinal interaction is not revealed.
Valatsiklovir is brought mainly with urine by active canalicular secretion. Drugs with the similar mechanism of removal, can increase concentration of a valatsiklovir in blood plasma.
Cimetidinum and probenetsid increase the area under a curve acyclovir concentration/time by decrease in its renal clearance, nevertheless need of dose adjustment is absent in view of the wide therapeutic index of acyclovir.
It is necessary to be careful, appointing Valavir in high doses (4 g/days) for prevention of a Cytomegaloviral infection along with drugs which compete with acyclovir for ways of removal as it can lead to increase in level in blood plasma of one or both drugs and their metabolites. At a concomitant use from the mikofenolat mofetily (the immunosuppressor drug which is applied at organ transplantation) in blood plasma the level of acyclovir and an inactive metabolite of the mikofenolat of a mofetil increases. It is necessary to appoint carefully Valavir in high doses (4 g and more) with other drugs which influence function of kidneys (for example cyclosporine, takrolimus).
Special instructions
Sick advanced age
For patients of advanced age in the absence of the significant changes of function of kidneys it is not recommended to change a dose. In a situation when renal failures are possible, the dose of drug needs to be adjusted.
It is necessary to support the adequate level of hydration of an organism.
Use in a renal failure (including at patients of advanced age)
Valatsiklovir is removed through kidneys, in this regard patients of advanced age with reduced function of kidneys need to reduce a drug dose. It is necessary to appoint carefully valatsiklovir the patient with a renal failure. It is obligatory to support the adequate level of hydration of an organism.
At patients with pathology of kidneys in the anamnesis the risk of development of neurologic complications increases therefore such patients demand careful observation. These reactions are in most cases reversible and disappear after drug withdrawal (see. Side effects).
To the patients who are on a hemodialysis, Valavir is recommended to apply in the same doses, as to patients with clearance of creatinine of ≤15 ml/min., the drug is taken after the procedure of a hemodialysis.
The clearance of creatinine has to be controlled constantly especially when function of kidneys can quickly change, for example right after transplantation. Respectively it is necessary to korrigirovat Valavir’s dose.
Dosing in an abnormal liver function
patients have no need for dose adjustment with cirrhosis of light or moderate expressiveness.
Use of higher doses of Valavir in a liver failure and transplantation of a liver
of Data on use of high doses of Valavir (≥4 mg/days) for treatment of patients with diseases of a liver is not present. Therefore it is necessary to appoint with care high doses of drug such patient. Special researches concerning Valavir’s use at transplantation of a liver were not conducted, however is established that prevention by means of high doses of a valatsiklovir reduces the frequency of infection and a disease of a Cytomegaloviral infection.
Reduction of transfer of a virus of genital herpes
of Supressivnaya therapy valatsikloviry reduces risk of transfer of genital herpes, but does not cure a herpes infection and completely does not exclude risk of transfer of a virus. In addition to therapy by Valavir it is recommended to follow the rules of safe sex.
Use in pediatrics
Is not present data on safety of use of drug for children up to 12 years.
There Is no feature of influence of medicine on ability to run vehicles and potentially dangerous mechanisms data on influence of drug on speed of response, but considering that sensitive patients at use of drug can have side reactions (dizziness, disturbance and confusion of consciousness, a hallucination, a loss of consciousness), such patients should refrain from control of vehicles and performance of other works demanding concentration of attention.
Overdose
Symptoms: nausea, vomiting. It was reported about cases of developing of an acute renal failure and neurologic symptoms (confusion of consciousness, a hallucination, agitation, a loss of consciousness and a coma) at patients at overdose valatsikloviry.
Many of the described cases of repeated overdose with renal failures and at patients of advanced age were caused in patients by an insufficient dose decline of drug.
Treatment: patients are subject to careful observation for timely diagnostics of toxic manifestations. The hemodialysis considerably accelerates removal of acyclovir from blood and can be considered as an optimum way of treatment.
The form of release and packing
of the Tablet coated on 500 mg.
On 6 or 10 tablets in blister strip packaging (blister).
On 7 planimetric packs (on 6 tablets) or on 1 planimetric packing (on 10 tablets) together with the instruction for medical use place in a pack from cardboard.
To Store storage conditions in the dry, protected from light place at a temperature not above 25 °C.
To store out of children’s reach!
A period of storage
2 years
it is not necessary to use drug after the termination of the expiration date specified on packing.
Prescription status
According to the prescription
JSC Pharmak Adres Producer: Ukraine, 04080, Kiev, st. of Frunze, 63.
The address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products (goods)
Republic of Kazakhstan, 050034 Almaty, Brodsky St., 37A, office 217,
Ph. / fax: +7 (727) 227 – 37 – 21 (22)
the E-mail address:
To Develop znamer@ukr.net
Additional information
| Ingredient |
|---|


















Reviews
There are no reviews yet.