Description
The instruction for medical use
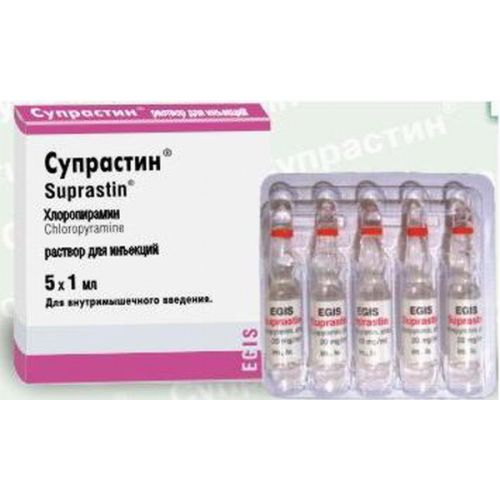
of SUPRASTIN® medicine
the Trade name
of Suprastin®
the International unlicensed
name Chloropyraminum Dosage Form Solution for injections
Structure
of 1 ml of solution contains
active agent – Chloropyraminum a hydrochloride of 20 mg,
excipient – water for injections.
The description
Transparent colourless aqueous solution with a slight characteristic smell
Pharmacotherapeutic group
Antihistaminic drugs of systemic action. The ethylene diamines substituted. Chloropyraminum
ATH R06AC03 Code
the Pharmacological
Pharmacokinetics Well properties is distributed in an organism, including the central nervous system. Linking with proteins of plasma – 7.9%. The peak of binding is noted at rn 7.4. It is removed mainly by kidneys in the form of metabolites. At children the removal of drug happens quicker, than at adult patients.
At patients with an abnormal liver function and kidneys metabolism of Chlorpyraminum goes down therefore there can be a need of lowering of a dose.
A pharmacodynamics
Chloropyraminum – the chlorinated analog of a tripelenamin (piribenzamin), the antihistaminic drug of the first generation belonging to group of etilendiaminovy antihistaminic drugs. Prevents development and facilitates a course of allergic reactions. Effect of Chloropyraminum is based on blockade of H1 receptors. Drug also has effect on smooth muscles, permeability of capillaries and the central nervous system. Has peripheral anticholinergic activity, moderate spasmolytic properties.
Indications
– allergic diseases, including urticaria, seasonal allergic rhinitis, allergic conjunctivitis, food and medicinal allergy,
– allergic reactions to stings of insects
– contact dermatitis, a dermatografizm
– adjuvant therapy of system anaphylactic reaction and a Quincke’s disease
the Route of administration and doses
For intramuscular introduction.
Adult: the recommended daily dose – 20-40mg (1-2ml) intramusculary.
To children:
Age
the Recommended initial doses
of 1 – 12 months:
5 mg (¼ ampoules – 0.25 ml of solution) intramusculary
1 – 6 years:
10 mg (½ ampoules – 0.5 ml of solution) intramusculary
6 – 14 years:
10-20mg (½ ampoules 0.5-1ml solution) intramusculary
Further therapy and/or increase in a dose substantially are defined by presence of side effects.
The maximum daily dose should not exceed 2 mg/kg of body weight.
At an acute anaphylaxis or acute heavy allergic reaction it is recommended to begin treatment with a careful slow intravenous injection then to continue intramuscular injections or administration of drug inside.
At patients of advanced age and the exhausted patients: use of Suprastina demands extra care as antihistaminic drugs cause side effects in these patients more often (dizziness, drowsiness, a lowering of arterial pressure).
In an abnormal liver function: the dose decline in connection with decrease in metabolism of active component of drug in liver diseases can be required.
In a renal failure: change of the mode of administration of drug and a dose decline can be required because active component is generally emitted through kidneys.
Treatment duration:
Duration of treatment depends on the nature of symptoms, time and their manifestation.
Opening of ampoules (for right-handed persons):
Take the ampoule body in the left hand, between the big and bent index fingers. The point of a break has to be above. You hold a head of an ampoule between the big and bent forefinger of the right hand so that the thumb was on a break point, covering it. Leaning on a forefinger of the left hand, press on an ampoule a thumb of the right hand, putting, thus, the constant pressure of average force. Do not bring closer and do not distance from each other the right and left hands. The neck of an ampoule can crack at any time after pressing that you can not feel.
Contraindications
– hypersensitivity to active or other auxiliary component of drug
– a bad attack of bronchial asthma
– a prostatauxe
– the period of a neonatality and prematurity
– pregnancy and the period of a lactation
Side effects
Seldom (≥ 1/10,000 and & lt, 1/1000)
– a leukopenia, an agranulocytosis, hemolytic anemia, other dyscrasias from the system of blood
It is very rare (& lt, 1/10,000)
– allergic reactions
– sedation, fatigue, dizziness, an ataxy, a tremor, nervous excitement, spasms, a headache, euphoria, encephalopathy
– increase in intraocular pressure, an attack of glaucoma, disturbance of visual acuity
– arterial hypotension, tachycardia, arrhythmia
– loss or increase in appetite, unpleasant feelings or pain in epigastric area, dryness in a mouth, nausea, vomiting, diarrhea, a constipation
– a photosensitization
– a myopathy
– the complicated urination, an ischuria
When prescribing drug it is necessary to consider a possibility of development of sedative side reactions.
Medicinal interactions
, for example, moklobemid or selegilin strengthen Inhibitors of a monoaminooxidase (MAO) and extend anticholinergic effects of Suprastina.
It is necessary to observe extra care at simultaneous use of Suprastina with anxiolytic sedatives, tranquilizers, opioid analgetika, tricyclic antidepressants, atropine and other muskarinergichesky parasympatholytics (Suprastin and any of these drugs can enhance effects of each other).
Anti-histamine drugs suppress skin reactions in response to allergichesy skin tests, it some days before conducting such skin tests it is necessary to cancel use of this drug.
During treatment it is necessary to exclude consumption of alcoholic beverages (alcohol enhances the oppressing effect of Suprastina on the central nervous system).
Special instructions
Due to presence of anticholinergic and sedative effects it is necessary to appoint with care Suprastin at patients of advanced age and also in an abnormal liver function, a cardiovascular system, in closed-angle glaucoma, an ischuria, a prostate hypertrophy.
Use of drug in late evening hours can strengthen symptoms of a gastro-ezofagalny reflux disease.
At a combination to the ototoksichesky drugs Suprastin can mask precursory symptoms of ototoxicity.
Prolonged use of Suprastina in rare instances can cause disturbances from the system of a hemopoiesis (leukopenia, an agranulocytosis, thrombocytopenia, hemolytic anemia).
At the developing of fever of not clear origin, laryngitis, an ulceration of a mucous membrane of a mouth, pallor, jaundice, hemorrhages unusual or difficult stopped bleedings, at long administration of drug it is necessary to define quantity of uniform elements of blood, and at detection of disturbances from the system of a hemopoiesis the administration of drug should be stopped.
Alcohol enhances sedation of anti-histamine drugs on the central nervous system, on it during treatment of Suprastinom® the consumption of alcoholic beverages is forbidden.
Pregnancy and the period of a lactation
Are absent data of the controlled clinical trials on use of drug during pregnancy. However at newborns whose mothers took the anti-histamine drugs last months of pregnancy cases of development of a retrolental fibroplasia were described.
Due to the lack of sufficient data drug should not be used when breastfeeding.
Features of influence on ability to run the vehicle and potentially dangerous mechanisms
Drug can cause, especially in an initial stage of treatment, drowsiness and disturbance of psychomotor functions. In initial, individually defined, the period of use of Suprastina driving and occupations is forbidden by other potentially dangerous types of activity demanding speed of psychomotor reactions. In the course of further treatment the extent of restrictions is defined individually in dependence of tolerance of drug.
Overdose
Symptoms: hallucinations, concern, ataxy, lacks of coordination of movements, athetosis, spasms. At children of early age the excitement prevails. Sometimes there is a dryness in a mouth, the fixed mydriatic pupils, a dermahemia of the person, sinus tachycardia, an ischuria, fever. At adults – fever and a dermahemia of the person can be absent, after the period of excitement spasms and a postconvulsive depression, with possible development of a coma and cardiorespiratory insufficiency which can lead to the death of the patient within 2-18 hours follow.
Treatment: monitoring of indicators of a respiratory, cardiovascular system, symptomatic therapy, according to indications resuscitation actions is recommended. Specific antidote is not known.
A form of release and packing
On 1 ml in ampoules of colourless neutral glass with a point of a break of white color. Apply on a nimbus of ampoules with indelible paint a code ring of dark red color.
On each ampoule paste the label from paper self-adhesive.
On 5 ampoules place in blister strip packaging from a film polyvinylchloride and films of PETF/PE.
On 1 blister strip packaging together with the instruction for medical use in the state and Russian languages place in a pack from cardboard.
To Store storage conditions at a temperature from 15 wasps up to 25 wasps.
To store out of children’s reach!
Not to use a period of storage of 5 years after an expiration date.
Prescription status
According to the prescription
BUDAPEST CJSC EGIS PHARMACEUTICAL PLANT Producer 1106, Keresturi St., 30-38 Hungary
Phone number: (36-1) 803-5555, the fax: (36-1) 803-5529
Owner of the registration certificate
of CJSC EGIS Pharmaceutical Plant, Hungary
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products (goods)
Representation in RK CJSC EGIS Pharmaceutical Plant
050060, Almaty, Zharokov St. of 286 G
ph. + 7 (727) 247 63 34, + 7 (727) 247 63 33, fax: + 7 (727) 247 61 41
e-mail:
To Develop egis@egis.kz
Additional information
| Ingredient |
|---|

















Reviews
There are no reviews yet.