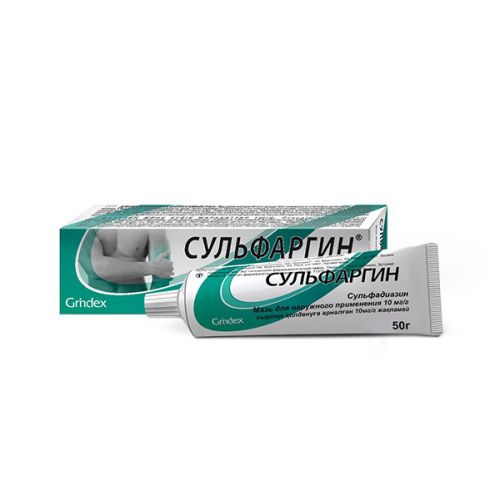
SULFARGIN® (Silver Sulfadiazine) Ointment 1% 50g tube
$22.00
Description
The instruction for medical use of SULFARGIN® medicine Trade name СУЛЬФАРГИН® the International unlicensed name Sulfadiazinum Dosage Form Ointment for external use of 10 mg/g Structure of 1 g of ointment contains active agent – Sulfadiazinum of silver of 10 mg, excipients: liquid paraffin, propylene glycol, cetostearyl alcohol, glyceryl monostearate 40-55, polysorbate 80, methylparahydroxybenzoate, propilparagidroksibenzoat, the water purified to 1 g. The description Ointment of white or almost white color with a characteristic smell. Pharmacotherapeutic group Antimicrobial drugs for topical administration. Streptocides. Silver Sulfadiazinum the ATX D06BA01 Code the Pharmacological Pharmacodynamics Sulfadiazinum properties of silver has antimicrobic effect on gram-positive and gram-negative microbes and mushrooms (Candida, Phycomycetes and Aspergillus spp, dermatophytes). The activity of drug is based on silver ions which are released in a wound as a result of moderate dissociation of Sulfadiazinum of silver, than long antimicrobic action which supplements Sulfadiazinum (streptocide) is reached. Ions of silver are adsorbed on a surface of microbic cells and render so-called oligodynamic bacteriostatic and bactericidal action. Drug does not possess nekrolitichesky and mutagen action. Ointment is characterized by moderate osmotic activity. The pharmacokinetics At topical administration can be soaked up to 1% of ions of silver and up to 10% of Sulfadiazinum of the total amount of ointment applied on a wound. The most part of the soaked-up silver is allocated with bile. Sulfadiazinum does not collect in an organism and is quickly emitted through kidneys. Indications – prevention and treatment of the infected burn wounds – the infected skin ulcers and decubituses (as a part of complex therapy) – prevention of infection of the replaced sites of skin and extensive grazes the Route of administration and doses Outwardly. After removal of necrotic fabrics, ointment is applied a thin layer (2 4 mm) on the damaged surface by 1-2 times a day under a sterile bandage or an open way. The maximum single dose – 300 g. Treatment duration – up to 3 weeks. At use of ointment on an extensive surface it is necessary to control function of kidneys and a liver and the maintenance of uniform elements of blood. The patient should give plentiful alkaline drink. Side effects Side effects are classified according to groups of systems and bodies and also frequency of occurrence of MedDRA: very often (≥1/10), it is frequent (≥1/100 to & lt, 1/10), is more rare (≥1/1000 to & lt, 1/100), it is rare (≥1/10,000 to & lt, 1/1000), is very rare (& lt, 1/10,000), it is unknown (it is impossible to determine by the available data). Local reactions from skin and hypodermic fabrics Seldom: allergic reactions (burning sensation, an itching, rashes) It is very rare: local irritative action in the form of short-term burning, pain which undergo System reactions in 5-10 minutes the Concentration of streptocide in blood plasma can reach the same level, as well as in case of system therapy if at a wound a big surface or treatment it is long. In such cases side effects, characteristic of streptocides, can be shown: From blood and lymphatic systems Seldom: a leukopenia, thrombocytopenia, an eosinophilia from mentality It is unknown: alarm from nervous system Seldom: headaches, spasms from digestive tract Often: nausea, vomiting, a diarrhea, a glossitis from a liver and/or a bile-excreting system Seldom: a liver failure from skin and hypodermic fabrics Seldom: an argiriya (as a result of accumulation of silver in fabrics, skin can become ash-gray). Usually appears at use of ointment on extensive surfaces or at prolonged use. From the skeletal and muscular and accompanying systems It is unknown: a joint pain from kidneys and an urinary system it is rare: a crystalluria, a renal failure inform the doctor On any manifestations of side effects. Contraindications – hypersensitivity to Sulfadiazinum of silver or any excipient of drug – hypersensitivity to streptocides – genetic deficit glyukozo-6-fosfatdegidrogenazy: at use on extensive surfaces there can be a hemolysis – early children’s age (newborns, up to 3 months), premature children in connection with danger of developing of a kernicterus – a porphyria – pregnancy, the lactation period – is inexpedient to apply to treatment of deep purulent wounds and burn wounds with plentiful exudation Medicinal interactions Sulfadiazinum of silver can inactivate fermental drugs for clarification of a wound therefore simultaneous use of these drugs is not recommended. Special instructions With care appoint drug in a liver and renal failure. If treatment is carried out it is long or apply drug on extensive burn surfaces, then in connection with danger of emergence of a leukopenia, thrombocytopenia or an eosinophilia should define quantity of leukocytes of blood regularly. Also as at topical administration of other drugs with antimicrobic action, at use of Sulfadiazinum of silver there can be superinfections. Sulfadiazinum of silver cannot apply use in pediatrics premature children and children aged up to 3 months in connection with danger of developing of a kernicterus. Pregnancy and the period of a lactation Data on safety of use of Sulfadiazinum of silver during pregnancy are absent therefore use of ointment during pregnancy and a lactation is contraindicated. Ointment contains in quality of excipients methylparahydroxybenzoate, propilparagidroksibenzoat which can cause allergic reactions (also slowed down type), in some cases – a bronchospasm. Ointment also contains cetostearyl alcohol which can cause local skin reactions (for example, contact dermatitis), and propylene glycol which can cause irritation of skin. Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms. Drug does not affect ability to run the vehicle and to work with technical devices. Overdose Symptoms. At long-term treatment of extensive burn surfaces perhaps systemic action of streptocides: nausea, vomiting, diarrhea, glossitis, joint pains, liver failure, headache, confusion of consciousness, convulsive spasms, crystalluria, renal failure, alarm, leukopenia, thrombocytopenia, eosinophilia. Symptomatic treatment. A form of release and packing On 50 g in tubas aluminum with the internal surface varnished with a protective aluminum membrane on a mouth and the cone-shaped device for a membrane puncture in screwing on to the bushena from plastic. The tuba together with the instruction for medical use in the state and Russian languages is placed in a pack from cardboard bandbox. To Store storage conditions at a temperature not above 25 ºC. Not to freeze! To store out of children’s reach! 2 years not to apply a period of storage after the expiration date specified on packing. Prescription status Without prescription JSC Grindex Producer. Krustpils St., 53, Riga, LV-1057, Latvia the Owner of the registration certificate of JSC Grindex, Latvia the Organization accepting claims for territories of RK Representative office of JSC Grindex 050010, Almaty, Dostyk Ave., a corner of Bogenbay St. of the batyr, 34a/87a, office No. 1
Additional information
| Ingredient |
|---|



















Reviews
There are no reviews yet.