Description
The instruction for medical use of medicine
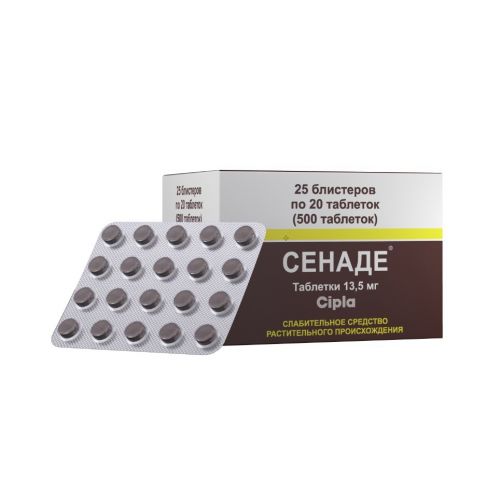
SENADUM (Senade)
the Trade name
Senadum
the International unlicensed name
Is not present
the Dosage form
of the Tablet of 13.5 mg
Structure
One tablet
calcium sennozid 15% contains active agent………………… 90.00 mg
(it is equivalent to 13.5 mg of Senna of calcic salts of sennozid And yes C),
excipients: lactose, starch corn, cellulose microcrystalline, the talc purified, methylparahydroxybenzoate, magnesium stearate, sodium lauryl sulfate, sodium of a karmelloz.
The description
of the Tablet of round shape, with a flat surface, brown or dark brown color, with the facet risky on one party and Cipla engraving on other party.
Pharmacotherapeutic group
Laxatives. Contact laxatives. Sennas glycosides.
ATH A06AB06 code
The pharmacological
Pharmacokinetics Active Ingredients properties of drug (metabolites of antraglikozid of Senna) are practically not soaked up, generally removed with a stake and insignificant quantity with urine. In a large intestine the bigger quantity of sennozid is hydrolyzed under the influence of enzymes of indestinal flora in pharmacological active free anthraquinones. System bioavailability of sennozid And & lsaquo, 5%. Laxative action comes in 8 – 10 hours after reception.
A pharmacodynamics
the Laxative effect is caused by reflex strengthening of a vermicular movement owing to impact on receptors of a large intestine. Products that are formed of senozid with participation of fermental systems of normal intestinal microflora, cause direct influence on receptors of a mucous membrane. Besides, drug promotes increase in volume of contents of intestines and accelerates motility of a large intestine. It is caused by suppression of absorption of ions of sodium, water, D-xylose and stimulation of secretion of sodium and water in an intestines gleam by increase in concentration of E2 prostaglandin in its walls.
Indications
– chronic constipations
– regulation of a chair in hemorrhoids, a proctitis, anal cracks
– preparation for X-ray inspections.
The route of administration and doses
Accept inside usually 1 time a day in the evening before going to bed,
Adults and children are more senior than 12 years: on 1 tablet on reception before going to bed, washing down with water or any drink. Maximum daily dose 3 tablets.
Children of 6-12 years: on & frac12, tablets before going to bed. Maximum daily dose 2 tablets.
In the course of selection the same dose should be accepted within several days and to gradually increase it on & frac12, tablets. If after achievement of the maximum dose within 3 days the defecation does not happen, it is necessary to see a doctor.
Side effects
Are possible:
– kolikoobrazny abdominal pain, a meteorism
– loss with a stake of electrolytes, generally potassium and disturbance of water and electrolytic exchange
– an albuminuria, a hamaturia, adjournment of melanin in a mucous membrane of intestines
– nausea, vomiting, diarrhea, urine decolouration, skin rash
– spasms, vascular collapse (at prolonged use, especially in high doses).
Contraindications
– hypersensitivity to glycosides of Senna and other components of drug
– a spastic constipation, intestinal impassability, an abdominal pain of not clear genesis
– the restrained hernia
– a round ulcer of a stomach and duodenum
– acute inflammatory bowel diseases and an abdominal cavity, peritonitis
– gastrointestinal bleedings,
– uterine bleedings
– cystitis
– disturbances of water and electrolytic exchange
– children’s age up to 6 years.
Medicinal interactions
At prolonged use Senadum in high doses strengthening of effect of cardiac glycosides and influence on effect of antiarrhytmic drugs in connection with a possibility of development of a hypopotassemia is possible.
At simultaneous use with thiazide diuretics, glucocorticoids, drugs of a root of a licorice the risk of development of a hypopotassemia increases. Breaks absorption of tetracyclines.
The special
instructions Drug should be taken with caution in diseases of a liver and/or kidneys, after band operations.
More than 2 weeks are not recommended to use drug.
After administration of drug, urine can gain yellowy-brown or red-lilac color.
Pregnancy and the period of a lactation
Drug should be taken with caution during pregnancy and feeding by a breast. The anthraquinone derivatives which are allocated with milk
cause diarrhea in children.
Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
Drug does not affect ability to drive the car and to work with the equipment. However it is necessary to check individual reaction to drug.
Overdose
Symptoms: the diarrhea leading to dehydration.
Treatment: conservative measures increase in consumption of liquid. It is usually enough to compensate loss of liquid and electrolytes. In certain cases it is necessary to make up for loss of liquid and electrolytes with the help in/in infusions of plasma substitutes.
The form of release and packing
On 20 tablets place in blister strip packaging from a film of the polyvinylchloride and printing aluminum foil varnished.
On 25 planimetric packs together with the instruction for medical use in the state and Russian languages place in a pack from cardboard.
To Store storage conditions in the dry place, at
a temperature not higher than 25 S. Hranit out of children’s reach!
A period of storage
3 years
not to apply after the expiration date specified on packing
Prescription status
Without prescription
Cipla Ltd producer. (Tsipla Ltd.), India
Meditab Spl.Pvt.Ltd., 352, Kundaim Indl.Estate, Kundaim, Goa, 403,115
Owner of the registration certificate
of Cipla Ltd. (Tsipla Ltd.), India
The address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products (goods)
of Kompaniya Avicenna Ltd in
Kazakhstan 050002, Almaty, Dzhangildin St. 31, business center Zhar-Su office 416
Ph. (8727) 357-23-87/88/89
E-mail: direct@avitsena.kz
Additional information
| Ingredient |
|---|



















Reviews
There are no reviews yet.