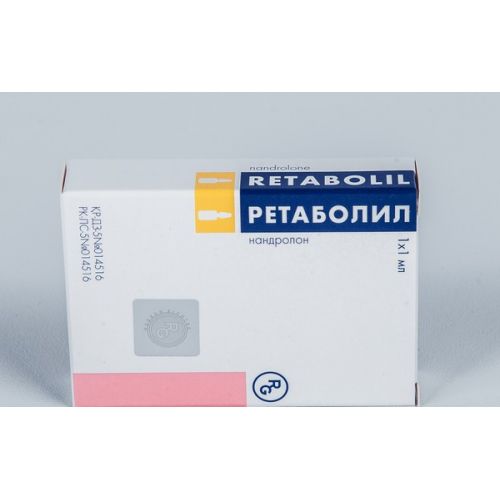
Retabolil 50 mg / ml solution for injection 1’s oil.
$17.10
Out of stock
Description
The instruction for medical use
of medicine
RETABOLIL
A trade name
Retabolil
the International unlicensed
name Nandrolonum Dosage Form
Solutio oleosa for injections
Structure
One ampoule of 1 ml contains
active agent – Nandrolonum of the dekanoat of 50 mg
excipients:
benzyl alcohol, isopropyl alcohol, sunflower oil for injections
the Description
the Yellow transparent Solutio oleosa free from particles.
The pharmacotherapeutic group
Anabolic steroid drugs – ethers
the Code of automatic telephone exchange A14AB01
the Pharmacological
Nandrolonum Pharmacokinetics properties dekanoat is slowly released from the place of an injection and gets to blood. Elimination half-life of 6 days. Ether will quickly be transformed to blood in Nandrolonum with elimination half-life ≤ 1 h. General time of hydrolysis of Nandrolonum of the dekanoat, distribution and removal of Nandrolonum of 4.3 h. Nandrolonum is metabolized in a liver. Nandrolonum metabolites – 19-norandrosteron, 19-noretiokholanolon and 19-norepiandrosteron, are defined in urine. Their pharmacological activity is unknown.
The pharmacodynamics
Retabolil is to synthetic derivatives of testosterone. Unlike testosterone possesses more significant anabolic and less significant androgenic action. Promotes synthesis of nucleic acids, proteins, causes a delay of nitrogen, calcium, sodium, potassium, chloride and phosphate. Activates reparative processes in an epithelium, bone and muscular fabrics.
Retabolil stimulates production of erythropoietin and stimulates an erythrogenesis, provides protection of marrow against the oppressing influence of cytotoxic agents that at simultaneous use of iron preparations is shown by antianemic action.
At observance of the recommended doses the androgenic effect (for example, virilescence) is observed seldom.
Indications
– a convalescence after severe injuries, intoxications, infectious diseases, extensive surgeries
– anemia as a result of chronic kidney disease (as a part of complex therapy)
the Route of administration and doses
Anemia:
weekly, intramusculary
– to women on 100 mg
– men on 200 mg.
The therapeutic effect has individual character. Treatment should be stopped in case of lack of therapeutic effect after 3-6 months of use.
After the corresponding improvement of a picture of blood or its normalization it is necessary to lower gradually a dose under regular control of hematologic indicators. Deterioration in indicators during a dose decline or after the end of treatment can serve as the indication to treatment resuming.
Convalescence:
on 50 mg each 3-4 weeks, intramusculary, it is deep in a muscle.
Side effects
– burning sensation in language, heartburn, a loss of appetite, nausea, vomiting
– a cholestasia, jaundice
– suppression of secretion of a gonadotrophin
– peripheral hypostases
– the strengthened skin vascularization
– a hypercalcemia
– virilescence at women (eels, a hirsutism, baldness on men’s type, irreversible decrease in a timbre of a voice, disturbance of a menstrual cycle, a clitoris hypertrophy)
– at men are suppression of function of testicles, an oligospermatism, a gynecomastia
– hypocoagulation with bent to bleedings
– delay or the termination of growth
– a hypertrophy and/or a carcinoma of a prostate
of the Contraindication
– children’s and teenage age up to 18 years
– pregnancy, the lactation period
– a hypertrophy or a carcinoma of a prostate
– a carcinoma of chest gland at men
– hypersensitivity to any of drug components
– a hydropic syndrome
Medicinal interactions
At co-administration with anti-diabetic means, indirect anticoagulants – coumarin derivatives, antiagregant it is necessary to show care because of possible strengthening of effect of these drugs.
At simultaneous use with glucocorticoids, mineralokortikoida, corticotropin, natriysoderzhashchy saline solutions, the food rich with sodium the liquid delay in an organism amplifies.
Special instructions
It is forbidden to exceed the recommended doses. When prescribing drug the women should weigh advantage and harm of treatment in view of androgenic effect of drug. Drug is appointed with care in dysfunction of a myocardium, a hypertension, an abnormal liver function and kidneys, epilepsy, migraine and glaucoma (including in the anamnesis). During treatment it is recommended to control intraocular pressure because of a possible delay in an organism of sodium and water.
Change of function of a liver (for example, the level of a bromsulftalein) is possible therefore after a 4 weeks course of treatment it is recommended to carry out control of its function. Before starting treatment and further, during it it is necessary to perform rectal examination of a prostate regularly. In diabetes it is necessary to reconsider dosages of anti-diabetic drugs.
Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
Effect of drug on the abilities necessary for control of motor transport and working cars, are not studied
Overdose
Symptoms: heavy endocrine, metabolic and mental side reactions develop.
Treatment: symptomatic.
A form of release and packing
of 1 ml of Solutio oleosa for injections in a colourless glass ampoule.
1 ampoule in a plastic form, in a cardboard box with the enclosed instruction for use.
To Store storage conditions at a temperature from +15 °C to +25 °C, in the place protected from light.
To store out of children’s reach!
Period of storage
of 5 years.
After expiry date not to use drug.
Prescription status
According to the prescription
Budapest JSC Gideon Richter producer 1103, Djemrei St., 19-21, Hungary
Additional information
| Ingredient |
|---|



















Reviews
There are no reviews yet.