Description
The instruction for medical use
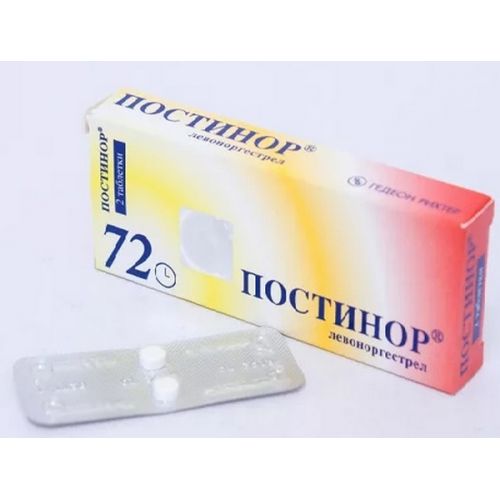
of POSTINOR® medicine
the Trade name
of Postinor®
the International unlicensed
name Levonorgestrel Dosage Form
of the Tablet of 0.75 mg
Structure
One tablet contains
active agent – levonogrestret 0.75 mg,
excipients: silicon dioxide colloidal anhydrous, potato starch, magnesium stearate, talc, starch corn, lactoses monohydrate.
The description
of the Tablet of round shape, with a flat surface, white or almost white color, with a facet, with marking of INOR on one party
Pharmacotherapeutic group
Sex hormones and modulators of a reproductive system. Hormonal contraceptives for system use. Progestogens. Levonorgestrel
the ATX G03AC03 Code
the Pharmacological
Pharmacokinetics At properties oral administration levonorgestrel is quickly and almost completely soaked up. In 1.6 hours after reception of one tablet Postinora® the maximum concentration of drug in blood serum reaches 14.1 ng/ml. After that there is two-stage lowering of concentration of drug, elimination half-life fluctuates ranging from 9 till 14.5 o’clock.
Levonorgestrel is equally removed with urine and a stake only in the form of metabolites. Biotransformation of levonorgestrel corresponds to metabolism of steroids. Levonorgestrel is hydroxylated in a liver and metabolites are removed in the form of the conjugated glyukuronat. Pharmacological active metabolites of levonorgestrel are unknown. Levonorgestrel contacts albumine of blood serum and the globulin, connecting sex hormones (G,CSH). Only 1.5% of all dose are in a free form, an of 65% is connected with GSPG. The absolute bioavailability is nearly 100% of all dose.
About 0.1% of a dose get to the child’s organism with maternal milk.
The pharmacodynamics
Levonorgestrel in the recommended dose prevents an ovulation and fertilization if sexual intercourse happened before an ovulation (at this time the probability of fertilization is higher). Besides, the endometrium changes caused by levonorgestrel, interfere with oospore implantation. If implantation process already began, then drug will be inefficient.
Efficiency: by means of the tablets Postinor® it is possible to prevent pregnancy approximately in 85% of cases. The more time passed between sexual intercourse and administration of drug, the lower it efficiency (95% within the first 24 hours, 85% – from 24 to 48 hours, and 58% – from 48 to 72 hours). Thus, reception of the tablets Postinor® is recommended to be begun as soon as possible (but no later than 72 hours) after sexual intercourse if no protective measures were taken. In the recommended dose levonorgestrel has no significant impact on the system of coagulation and on metabolism of lipid and carbohydrates.
Indications
– emergency contraception for the purpose of contraception (in the first 72 hours after sexual intercourse) in the absence of contraception or insufficient reliability of the applied
Route of administration method and a dose
Drug is used orally. It is necessary to take 2 pill. For achievement of more reliable effect it is necessary to take the first pill as soon as possible after sexual intercourse (not later, than in 72 hours). It is necessary to take the second pill in 12 hours (but not later, than in 16 hours) after reception of the first tablet.
If within three hours after reception of any tablet there was vomiting, then it is necessary to take one more pill.
The tablets Postinor® can be applied in any day of a menstrual cycle, but only provided that the previous periods were normal.
After intake of the emergency contraceptive to the following periods it is necessary to apply local barrier contraceptive methods (for example, condom). After use of the tablets Postinor®, constant use of oral contraceptives is not contraindicated.
Side effects
Very often (& gt, 1/10)
– nausea, pain in the lower part of a stomach
– the bleeding which is not connected with periods
– fatigue
Often (from & gt, 1/100 to & lt, 1/10)
– a headache, dizziness
– vomiting, diarrhea
– morbidity at a palpation of mammary glands
– a periods delay more than for 7 days
– irregular bleedings and bloody discharges
the Nature of periods can temporarily change, but at most of women the following monthly arrives on time, within 7 days since the expected moment. If the periods are late more than for 5 days, then it is necessary to exclude a possibility of pregnancy.
Contraindications
– the hypersensitivity to drug components
– girls till the puberty period
– patients
Medicinal interactions
– at a concomitant use of drugs inductors of enzymes of a liver happens to rare hereditary diseases of intolerance of a galactose, deficiency of Lappa lactase or a syndrome of glyukozo-galaktozny malabsorption levonorgestrel metabolism acceleration
– reduce efficiency of levonorgestrel: barbiturates, including Primidonum, Phenytoinum and carbamazepine, drugs containing a St. John’s wort (Hypericum perforatum) and also rifampicin, ritonavir, rifabutin, griseofulvin. The women taking these drugs should see a doctor
– the drugs containing levonorgestrel can increase risk of toxicity of cyclosporine, owing to suppression of his metabolism.
The special
instructions Contraceptive should be applied only in the emergency situations. Drug does not replace regularly applied contraceptives.
The emergency contraceptive can not always prevent pregnancy approach.
The efficiency of the tablets Postinor® after sexual intercourse at which contraceptives were not applied decreases over time:
Time between sexual intercourse and reception of the tablets Postinor®:
Efficiency:
Within 24 hours
of 95%
25-48 hours
of 85%
49-72 hours
of 58%
If there are doubts concerning time of the unprotected sexual contact or if the unprotected contact took place more than in 72 hours during the same menstrual cycle, then there is a probability that conception already happened. In this regard use of the drug Postinor® at repeated sexual contact can be inefficient concerning prevention of approach of pregnancy. If the menstrual cycle is late more than for 5 days or if in day of the expected periods the unusual bleeding takes place or if there are other bases to assume pregnancy approach – it is necessary to exclude pregnancy.
If pregnancy occurred after drug Postinor® use, it is necessary to consider a possibility of existence of ectopic pregnancy. Especially existence of ectopic pregnancy can be suspected at women who note severe pains in a stomach or unconscious states and also in the presence in the anamnesis of ectopic pregnancy, uterine tube surgery or inflammatory diseases of bodies of a small pelvis.
Use of drug is not recommended at patients with serious violation of function of a liver.
Severe forms of a sprue can have negative impact on efficiency of the drug Postinor® (for example, Crohn’s disease). The women having similar diseases in need of carrying out emergency contraception have to see a doctor.
This medicine contains lactoses monohydrate therefore it should not be applied to patients with rare hereditary diseases of intolerance of a galactose, deficiency of Lappa lactase or a syndrome of glyukozo-galaktozny malabsorption.
After administration of drug of Postinor® the periods usually pass normally and come in time. The periods can sometimes begin for several days earlier or later. Women should recommend to visit the doctor to pick up and begin to use one of methods of regular contraception. If in the following cycle after use of the drug Postinor® and regular hormonal contraception, the periods do not come, then pregnancy has to be excluded.
Repeated use of drug during one menstrual cycle is not recommended in connection with a possibility of disturbance of a cycle. Постинор® it is inefficient as a standard regular method of contraception and it can be used only as an emergency measure. Women who address concerning repeated courses of emergency contraception should recommend to use long-term methods of contraception.
Use of emergency contraception does not replace the necessary precautionary measures connected with protection against diseases sexually transmitted.
Use of the tablets Postinor® is not recommended at puberal girls up to 16 years without medical control.
Pregnancy and a lactation
of the Tablet Postinor® it is impossible to apply during pregnancy as this drug cannot interrupt the available pregnancy. According to epidemiological researches, adverse effect of progestogens on a fruit (was not revealed at accidental use of the emergency method of contraception).
Levonorgestrel gets into maternal milk. Potential impact of drug on the child can be reduced sharply if mother takes both pill directly after feeding, and after reception of tablets abstains from feeding.
Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
Impact of drug on ability to driving or works with mechanisms was not studied.
The overdose
Is not present data on development of heavy undesirable effects after reception of high doses of oral contraceptive drug.
Symptoms: nausea, breakthrough bleeding.
Treatment: symptomatic. Drug has no specific antidote.
The form of release and packing
On 2 tablets place in blister strip packaging from a film of polyvinylchloride and aluminum foil.
On 1 blister strip packaging together with the instruction for medical use in the state and Russian languages put
Storage conditions in a cardboard box
to Store at a temperature from 15 of 0C up to 25 0C.
To store out of children’s reach!
Not to use a period of storage of 5 years after expiry date drug.
Prescription status
According to the prescription
the Name and the country
of the JSC Gideon Richter manufacturing organization,
1103 Budapest, Dyomryoi St., 19-21, Hungary
the Name and the country of the owner of the registration certificate
of JSC Gideon Richter, Hungary
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products: Representative office of JSC Gideon Richter in RK E-mail: info@richter.kz Phone number: 8-(727) 258-26-22, 8-(727) 258-26-23
To develop
Additional information
| Ingredient |
|---|

















Reviews
There are no reviews yet.