Description
The instruction for medical use
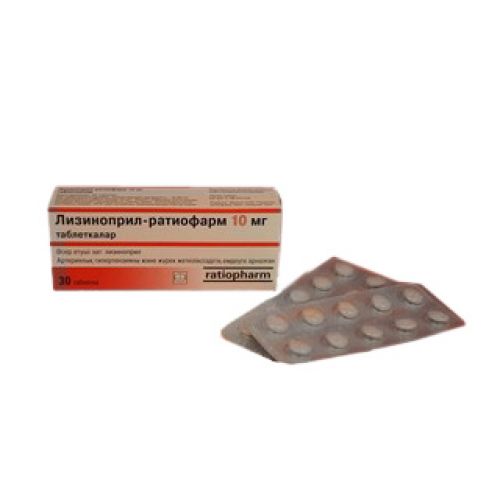
of Lizinopril-ratiopharm medicine of 5 mg, 10 mg, 20 mg
the Trade name
of Lizinopril-ratiopharm of 5 mg, 10 mg, 20 mg
the International unlicensed
name Lisinopril Dosage Form
of the Tablet on 5 mg, 10 mg, 20 mg
Structure
One tablet contains
active agent – lisinopril a dihydrate of 5.44 mg, 10.89 mg or 21.78 mg, is equivalent to lisinopril of anhydrous 5 mg, 10 mg, 20 mg,
excipients: Mannitolum, hydrophosphate calcium dihydrate, starch prezhelatinizirovanny, RV-24823 dye, sodium of a kroskarmelloz, magnesium stearate.
The description
of the Tablet of white color, round, biconvex, with risky on one party (for a dosage of 5 mg).
Tablets of light pink color, round, biconvex, with risky on one party (for a dosage of 10 mg).
Tablets of pink color, round, biconvex with risky on one party (for a dosage of 20 mg).
Pharmacotherapeutic group
the Drugs vliyashchy on a system renin-angiotensin. Enzyme Angiotensin-converting (EAC) inhibitors. Lisinopril.
The ATX C09AA03 code
the Pharmacological
Pharmacokinetics Maximum Concentration properties in blood plasma is reached approximately in 7 h after oral reception. Meal does not influence lisinopril absorption speed. Lisinopril does not contact proteins of blood plasma. The absorbed biologically active agent completely and in an invariable look is emitted through kidneys. The effective half-life period was 12.6 hours. Lisinopril gets through a placenta.
The pharmacodynamics
of Lizinopril-ratiopharm is inhibitor of angiotensin-converting enzyme (iAPF). Suppression of APF leads to the reduced formation of angiotensin II (the possessing vasoconstrictive action) and to decrease in secretion of Aldosteronum. Lizinopril-ratiopharm also blocks bradykinin disintegration – powerful vasodepressor peptide. Therefore it reduces arterial blood pressure, the general peripheric vascular resistance, before – and an afterload on heart, increases the minute volume, warm emission and increases tolerance of a myocardium to loadings and improves blood supply of an ischemic myocardium. At patients with an acute myocardial infarction lisinopril-ratiopharm together with nitrates reduces forming of dysfunction of a left ventricle or heart failure.
Indications
– arterial hypertension
– chronic heart failure (as a part of complex therapy with diuretics and cardiac glycosides)
– an acute myocardial infarction at patients with a stable hemodynamics without symptoms of dysfunction of kidneys.
Route of administration and dosage
Arterial hypertension:
Treatment has to begin with 5 mg daily in the mornings. The dose has to be established so that to provide optimum control of level of arterial blood pressure. Time interval between increases in a dose has to make not less than 3 weeks. The usual maintenance dose makes 10–20 mg of lisinopril once a day, a makimalny daily dose – 40 mg once a day.
Heart failure:
Lizinopril-ratiopharm it is appointed in addition to the available therapy diuretics and a digitalis. The initial dose makes 2.5 mg of lisinopril-ratiopharm in the mornings. The maintenance dose has to be established step by step with increases on 2.5 mg at an interval of 2–4 weeks. The usual maintenance dose makes 5–20 mg once a day. It is impossible to exceed the maximum dose of 35 mg lisinopril/day.
An acute myocardial infarction at patients with a stable hemodynamics:
Treatment by lisinopril-ratiopharm can begin within 24 hours after emergence of symptoms on condition of a stable hemodynamics (systolic pressure more than 100 mm Hg., without symptoms of dysfunction of kidneys), in addition to standard therapy of a heart attack (thrombolytic means, acetylsalicylic acid, beta blockers, nitrates). The initial dose makes 5 mg of lisinopril-ratiopharm, in 24 hours – 5 more mg, in 48 hours – 10 mg of lisinopril. Then the dose makes 10 mg of lisinopril once a day.
Patients with low systolic pressure (≤ 120 mm rt. stations) before therapy or during the first 3 days after a heart attack have to receive for treatment lower therapeutic dose – 2.5 mg of lisinopril-ratiopharm. If systolic pressure is less than 90 mm Hg. more than 1 hour it is necessary to refuse lisinopril-ratiopharm.
Treatment has to continue within 6 weeks. The minimum maintenance dose makes 5 mg of lisinopril-ratiopharm a day. Patients with symptoms of heart failure have to be treated by lisinopril-ratiopharm further. Lizinopril-ratiopharm it can be given along with nitroglycerine (intravenously or in the form of a skin plaster).
In a myocardial infarction lisinopril has to be given in addition to usual standard therapy (thrombolytic means, acetylsalicylic acid, beta blockers), it is preferable together with nitrates.
Elderly patients:
At elderly patients the dosage has to be corrected taking into account creatinine level (for assessment of function of kidneys) which is calculated by a formula Kokrofta:
Clearance of creatinine =
(140 – age) × the body weight (kg)
0.814 × concentration of creatinine in serum (µmol/l)
(For women the received result on this formula needs to be increased by 0.85).
A dosage at patients with moderately bounded function of kidneys (clearance of creatinine of 30 – 70 ml/min.):
The initial dose makes 2.5 mg of lisinopril-ratiopharm in the mornings, supporting – 5–10 mg a day. It is impossible to exceed the maximum dose of 20 mg of lisinopril a day.
Lizinopril-ratiopharm can be accepted irrespective of receptions of a pishcha, but with enough liquid, once a day, preferably at the same time.
Side effects
Sometimes
– headaches, dizziness, feeling of weakness, a disorder of vision, fatigue
– dry cough, a sore throat
– nausea, loss of appetite, diarrhea, a constipation, vomiting
– allergic rash
Seldom
– a consciousness oglushennost, a depression, sleep disorders, sonitus, disturbances of sense of equilibrium, nervousness, changes of taste
– a peripheral neuropathy with paresthesias, muscular spasms, inflow, sweating
– short wind, a bronchospasm, an allergic alveolitis
– tachycardia, disturbances of a warm rhythm, cardiogenic shock, retrosternal pains, orthostatic hypotension
– cholestatic jaundice, a proteinuria and increase in bilirubin and enzymes of a liver in blood serum
– anemia, thrombocytopenia, a neutropenia, an eosinophilia, in some cases an agranulocytosis or a pancytopenia, increase in concentration of urea, creatinine and potassium and decrease in concentration of sodium in blood serum (at patients with renal failures)
– in some cases: hemolytic anemias at patients with a considerable lack of glucose-3-phosphate-dehydrogenase
– a skin itching, a small tortoiseshell, a Quincke’s disease, a bladderwort, a multiformny erythema, exfoliative dermatitis, Stephens-Johnson’s syndrome, toxic epidermal necrosis, psoriasis, a photosensitization, an alopecia, an onycholysis
– Reynaud’s syndrome
– impotence
Very seldom
–
the Contraindication arthralgia
– the increased susceptibility to active agent or other iAPF or one of excipients
– a stenosis of renal arteries (bilateral or unilateral with one kidney)
– a Quincke’s disease in the anamnesis owing to former therapy iAPF and a hereditary/spontaneous Quincke’s disease
– heavy renal failures (clearance of creatinine & lt, 30 ml/min.).
– hemodynamically relevant stenosis of an aorta or the mitral valve or a hypertrophic cardiomyopathy
– an acute myocardial infarction at patients with an unstable hemodynamics
– systolic pressure ≤ 100 mm Hg. prior to treatment
– feeding by a breast
– pregnancy and the period of a lactation
– cardiogenic shock
– children’s age up to 18 years
– primary hyper aldosteronism
– a state after transplantation of a kidney
Medicinal interactions
At simultaneous use of the tablets Lizinopril-ratiopharm and:
– lithium lithium discharge from an organism can be reduced therefore it is necessary to watch attentively concentration of lithium in blood serum
– analgetics, non-steroidal anti-inflammatory drugs (for example, acetylsalicylic acid, indometacin) – easing of hypotensive effect of lisinopril
– Baclofenum – possibly strengthening of hypotensive effect of lisinopril – diuretics – possibly strengthening of hypotensive effect of lisinopril
– kaliysberegayushchy diuretics (Spironolactonum, Triamterenum or amiloride) is possible and potassium additives risk of development of a hyperpotassemia
– hypotensive drugs – possibly strengthening of hypotensive effect of lisinopril
– anesthetics, drugs, sleeping medicines – perhaps sharp lowering of blood pressure
– Allopyrinolum, tsitostatik, immunodepressants, system corticosteroids increases, procaineamide – risk of development of a leukopenia
– oral antidiabetic drugs (sulphonylurea derivatives, biguanides) increases and insulin – strengthening of hypotensive effect, especially in the first weeks of combination therapy is possible.
– an amifostina – strengthening of hypotensive effect
– antiacid means – reduction of bioavailability of lisinopril
– sympathomimetics – possibly strengthening of hypotensive effect
– alcohol – possibly strengthening of effect of alcohol
– table salt – weakening of hypotensive effect of lisinopril and emergence of symptoms of heart failure is possible.
Special instructions
to the Patients receiving several diuretics or diuretics in a high dosage (& gt, 80 mg furosemide/day), to patients with the hypovolemia, a hyponatremia (sodium content in serum & lt, 130 mmol/l) existing before the hypotension, unstable heart failure bounded by function of kidneys, high-dosage therapy by vasodilators and to patients at the age of 70 years are also more senior, it is recommended to begin therapy of Lizinoprilom-ratiopharm permanently.
At development of hypotension the strict medical observation, intake of diuretics in low doses and careful grinding of a dose of medicine in powder is necessary at simultaneous control of function of kidneys and level of potassium. If it is possible, it is necessary to stop temporarily therapy by diuretics in 2-3 days prior to therapy by lisinopril. It also should be taken into account to patients with stenocardia or other cardiovascular diseases in which because of excessive lowering of blood pressure the myocardial infarction or a stroke can develop.
At development of hypotension of the patient it is necessary to put on a back, if necessary carry out compensation of volume of the lost liquid. In bradycardia atropine is appointed.
Hemodialysis / LPNP-lipidny-aferez / Therapy for desensitization
At simultaneous use of lisinopril and dialysis about polyacrylic – a nitrile membrane or LDL (lipoprotein of low density) – an afereza by means of sulfate of a dextran or desensitization against poisons of insects (bees, wasps) the development of an acute anaphylaxis is possible.
It is recommended to use other membrane for dialysis or it is necessary to replace temporarily lisinopril-ratiopharm with other antihypertensive drugs (not iAPF).
A Quincke’s disease
At development of an anginevrotichesky face edema, lips antihistamines are appointed. Hypostasis of language, a glottis and/or throat can threaten life therefore the emergency treatment is carried out, with an immediate subcutaneous injection of 0.3-0.5 mg of epinephrine or slow intravenous input of 0.1 mg of epinephrine (observe instructions on dilution), with monitoring of the ECG and blood pressure.
The feature of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
needs to control the level of arterial blood pressure since. in connection with specific features at some patients the development of hypotension and, as a result, disturbance of ability to run motor transport and to serve moving mechanisms is possible.
Overdose
Symptoms: heavy hypotonia, shock, bradycardia.
Treatment: the patient has to be under strict medical observation, it is preferable in intensive care unit. It is necessary to control often concentration of electrolytes and creatinine in blood serum. The actions interfering absorption, such as gastric lavage, prescribing of adsorbents and sodium sulfate, have to be undertaken within 30 minutes after administration of drug. Lisinopril is removed a hemodialysis.
A form of release and packing
On 10 tablets in blister strip packaging from a film of PVC and aluminum foil.
On 3 or 5 planimetric packs together with the instruction for use in the state and Russian languages place in a pack from cardboard.
To Store storage conditions at a temperature not above 25ºС.
To store out of children’s reach!
4 years
not to apply a period of storage after an expiration date.
Prescription status
According to the prescription
Merkle GmbH Producer, Germany
the Owner of the registration certificate
“ratiopharm GmbH”, Germany
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products (goods) of ratiopharm Kazakhstan LLP, Almaty, Al-Farabi Ave., 19, BC Nurly-Tau, 1B, office 603, Phone number: (727) 3110915, Fax: (727) 3110734, e-mail:
To Develop teva@teva.kz
Additional information
| Ingredient |
|---|















Reviews
There are no reviews yet.