Description
The instruction for medical use
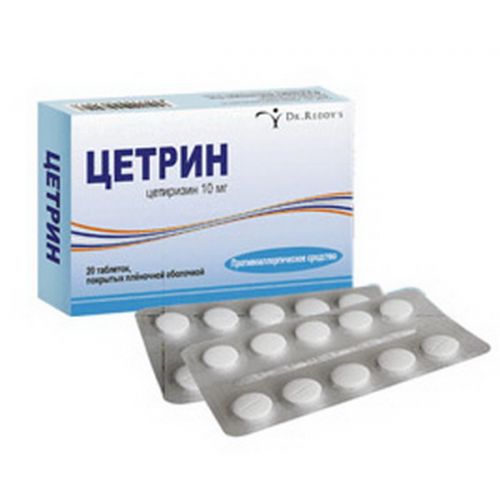
of Tsetrin® medicine
the Trade name
of Tsetrin®
the International unlicensed
name Tsetirizin Lekarstvennaya
the Tablet form, film coated 10 mg
Structure
One tablet contains
active agent – a tsetirizin a hydrochloride of 10 mg
excipients: lactose (450 mesh), starch corn, povidone (K-30), magnesium stearate
structure of a cover – a gipromelloz (hydroksipropilmetiltsellyuloza of 6 cps), sorbic acid, the titan E171 dioxide, talc purified, a macrogoal (Polyethyleneglycol 6000), polysorbate 80, dimetikon.
The description
of the Tablet of round shape, with a biconvex surface, film coated white color, with the line of a break on one party and a smooth surface on other party, diameter (8.20 ± 0.20) mm and thickness (3.50 ± 0.20) mm.
Pharmacotherapeutic group
Antihistaminic drugs of systemic action. Piperazin derivatives.
The code of automatic telephone exchange R06AE07
the Pharmacological
Pharmacokinetics Later properties of administration of drug inside the maximum concentration (Cmax) of a tsetirizin in plasma is reached within 1 hour. Administration of drug with food does not influence absorption size, but absorption speed
decreases a little.
Linking with proteins of plasma makes 93% and does not change at concentration of a tsetirizin in the range of 25 – 1000 ng/ml.
Pharmacokinetic parameters of a tsetirizin change linearly when assigning it in a dose of 5 – 60 mg.
Distribution volume – 0.5 l/kg. Gets into breast milk.
In small amounts it is metabolized in a liver by O-dealkylation with formation pharmacological of an inactive metabolite (unlike other blockers of the H1-histamine receptors which are metabolized in a liver with participation of a system of P450 cytochrome).
The system clearance is 53ml/min. Elimination half-life at adults makes 7 – 10 hours, children have 6 – 12 years – 6 hours, 2 – 6 years – 5 hours, 0.5 – 2 – 3.1 hours. At elderly patients the elimination half-life increases by 50%, the system clearance decreases by 40% (depression of function of kidneys).
Does not kumulirut. ⅔ drug also about 10% are removed in not changed look by kidneys – with stool. Is not removed at a hemodialysis.
The pharmacodynamics
Tsetrin – the selection antagonist of peripheral histamine H1 receptors, the carboxylated Hydroxyzinum metabolite, blocks H1-histamine receptors, drug of the prolonged action. Has antiallergic effect, inhibiting a late phase of migration of the cells participating in inflammatory reaction (generally eosinophils). Reduces an expression of molecules of adhesion, such as ICAM-1 and VCAM-2 which are markers of allergic inflammation.
Has insignificant anticholinergic and antiserotoninovy activity.
Citrine suppresses skin reactions at patients with a cold urticaria and a dermographism.
Indications
– seasonal and year-round rhinitis
– allergic conjunctivitis
– urticaria
– hay fever
– allergic dermatitis
– a Quincke’s disease
– atopic bronchial asthma as a part of complex therapy
the Route of administration and doses
Tsetrin is appointed inside, tablets wash down 200 ml of water, irrespective of meal.
To adults and children 6 years are more senior 2 times a day are appointed once on 10 mg or on 5 mg.
The maximum daily dose – 10 mg. The recommended daily dose cannot be exceeded.
Duration of treatment is defined individually and usually is 2 – 3 weeks. In seasonal allergic rhinitis – 3 – 4 weeks, at short-term influence of pollen – 1 week.
Side effects
– dryness in a mouth, nausea, vomiting, dyspepsia, an abdominal pain, diarrhea,
– drowsiness, fatigue, dizziness, a headache, migraine, decrease in flavoring sensitivity, irritability, excitability, a depression, aggressive reactions, spasms, hallucinations, a still birth
– pharyngitis, laryngitis, nasal bleeding
– a Quincke’s disease, rash, a small tortoiseshell, an itching
– a bronchospasm, cough
– tachycardia
– ALT/nuclear heating plant, alkaline phosphatase, bilirubin
– disturbance of urination
of the Contraindication
– hypersensitivity to any component of drug
– pregnancy and the period of a lactation
– children’s age up to 6 years
– heavy degree of chronic kidney disease
Medicinal interactions
of Pharmacokinetic interaction with pseudoephedrine, Cimetidinum, ketokonazoly, erythromycin, azithromycin, diazepam and glipizidy is not revealed.
Joint appointment with theophylline (400 mg/days) led to 16% to decrease in the general clearance of a tsetirizin (the kinetics of theophylline did not change).
Special instructions
Use for patients from the liver broken by function
to Adults and children are more senior than 12 years from the liver broken by function a daily dose of drug it is possible to lower to 5 mg once a day. Also at patients aged from 6 up to 11 years the recommended daily dose makes 5 mg once a day. Patients aged from 2 up to 5 years from the liver broken by function are not recommended to appoint tsetirizin.
Use for patients with impaired renal function
by the Adult and to children is more senior than 12 years with reduced function of kidneys or the patients who are on a hemodialysis a daily dose of drug can lower to 5 mg once a day. Also sick aged from 6 up to 11 years with impaired renal function the daily dose – 5 mg is recommended once a day. Patients aged from 2 up to 5 years with impaired renal function are not recommended to appoint tsetirizin.
Use for patients of advanced age
For patients of advanced age function of kidneys is often broken. Therefore further the dose is selected individually and perhaps there will be a need for control of function of kidneys.
At a concomitant use with alcohol the emergence of feeling of alarm and decrease of the activity of central nervous system is possible.
Features of influence of medicine on ability to run the vehicle or potentially dangerous mechanisms
With care appoint drug to the patients who are engaged in potentially dangerous types of activity requiring special attention and speed of psychomotor reactions as drug can cause drowsiness.
Overdose
Symptoms: drowsiness, concern, acrimony, an ischuria, tachycardia, dryness in a mouth, zaporoshok
Treatment: drug withdrawal, gastric lavage, intake of activated carbon, carry out symptomatic therapy.
The form of release and packing
On 10 tablets place in blister strip packaging from a film of the polyvinylchloride and printing aluminum foil varnished.
On 2 blister strip packagings together with the instruction for medical use in the state and Russian languages place in a pack from cardboard bandbox.
On 5 packs place in group packing.
Storage conditions
In the dry, protected from light place at a temperature not above 25 °C. To store in the places inaccessible for children!
2 years
not to apply a period of storage after the expiry date specified on packing.
Prescription status
According to the prescription
Dr. of Reddi’s Laboratoris Limited Producer
7-1-27 Amerpert, Hayderabad-500,016, India
the Owner of the registration certificate
Dr. of Reddi’s Laboratoris Limited, India
the Address of the organization accepting in the territory of the Republic of Kazakhstan claims from consumers on quality of products
Representative office Dr. of Reddi’s Laboratoris Limited in the Republic of Kazakhstan:
050057 Almaty, 22nd Line St., 45, 8 (727) 3941699, 8 (727) 3941689, fax: 3941294 (110), info@drreddys.kz
Additional information
| Ingredient |
|---|
















Reviews
There are no reviews yet.